A well-maintained lawn can help beautify our environment, reduce water runoff, and reduce water and air pollution. In 1992 there were 3.3 million acres of grass grown as home lawns in Florida, making home lawns the largest acreage crop in the sunshine state. For grass, just like any other crop, pest management is crucial for proper plant health. Plant-parasitic nematodes (Figure 1) are among the least understood and most difficult pests to manage on turfgrass in Florida.
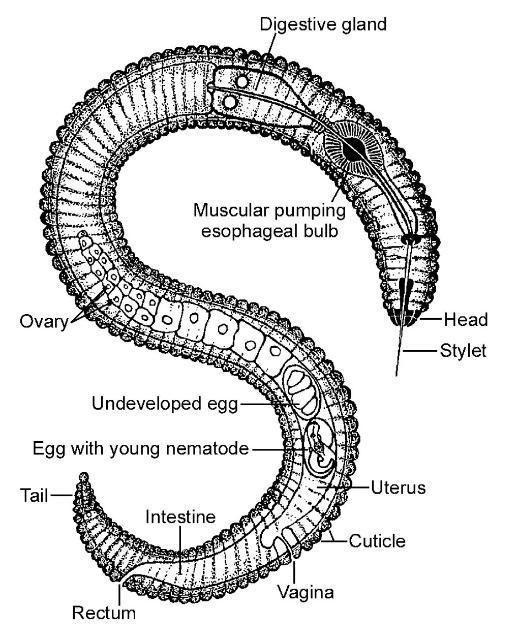
Credit: R. P. Esser, Florida Department of Agriculture and Consumer Services, Division of Plant Industry; used with permission.
Plant-Parasitic Nematodes
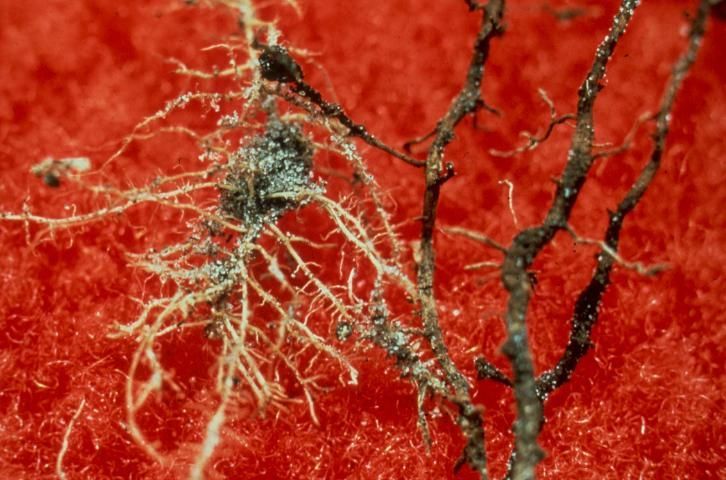
Nematodes are unsegmented roundworms, different from earthworms, flatworms, and other worms that are more easily observed. Nematodes living in soil are very small, and most can only be seen with the aid of a microscope (Figure 2). Many kinds of nematodes live in the soil in a home lawn. Most soil nematodes are beneficial, feeding on bacteria, fungi, or other microscopic organisms found in soil. In addition, some nematodes can be used as biological control organisms to help manage important insect pests. Unfortunately, other nematodes are harmful to plants because they feed on plant tissues and cause plant damage.
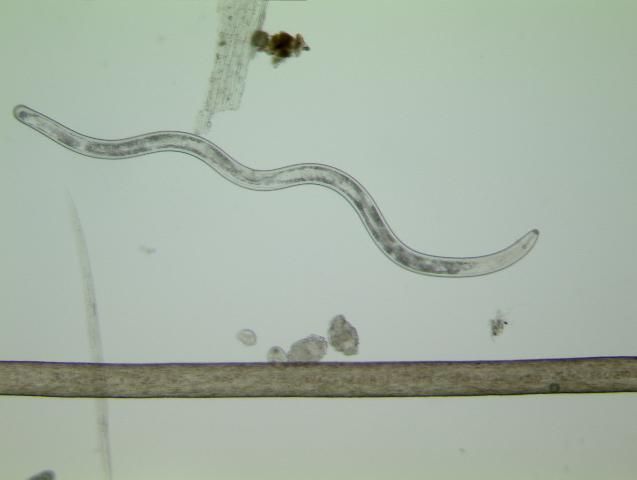
Credit: W. T. Crow, UF/IFAS
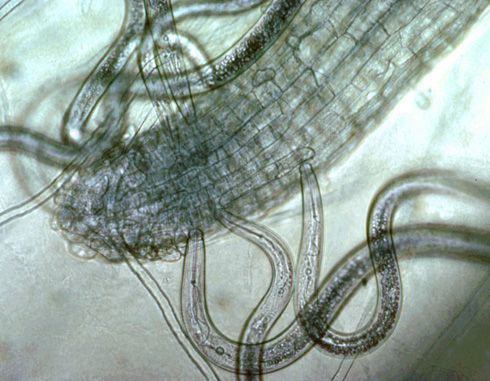
All plant-parasitic nematodes have a stylet or "mouth-spear" that is similar in structure and function to a hypodermic needle (Figure 3a and 3b). The nematode uses its stylet to puncture plant cells, inject digestive juices, and ingest plant fluids. All plant-parasitic nematodes that are important turfgrass pests in Florida feed on roots. Some plant-parasitic nematodes remain in the soil and feed by inserting their stylet into the root. These are called ectoparasitic nematodes (Figure 4). Others, using their stylet to puncture an entry hole in the root, feed with their body inside the root tissue. These are called endoparasitic nematodes (Figure 5). Of the common nematodes that cause damage to turfgrasses in Florida, sting, awl, stubby-root, sheath, sheathoid, spiral, and ring nematodes are strictly ectoparasites. Root-knot, lance, and lesion nematodes are endoparasites.
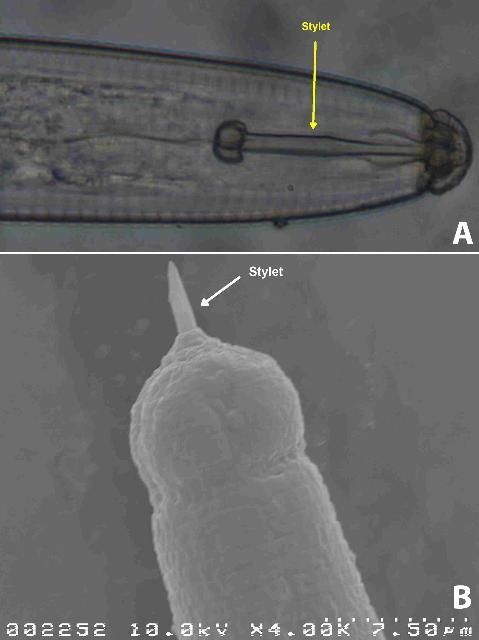
Credit: J. E. Luc, UF/IFAS
Credit: J. O. Becker, UC Riverside
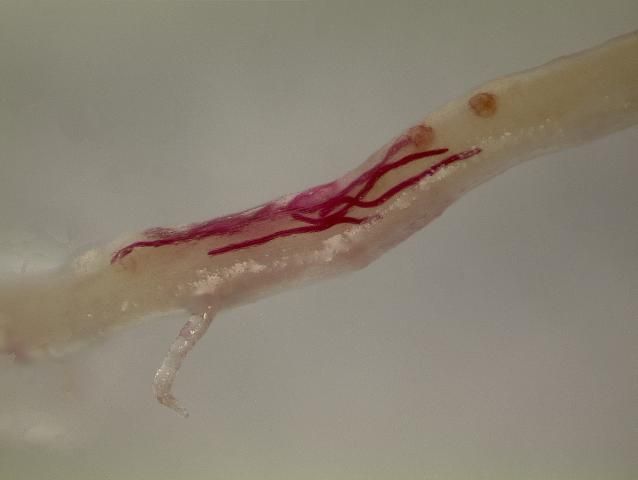
Credit: A. C. Hixson, UF/IFAS
Effects and Symptoms of Nematodes on Lawn Grasses
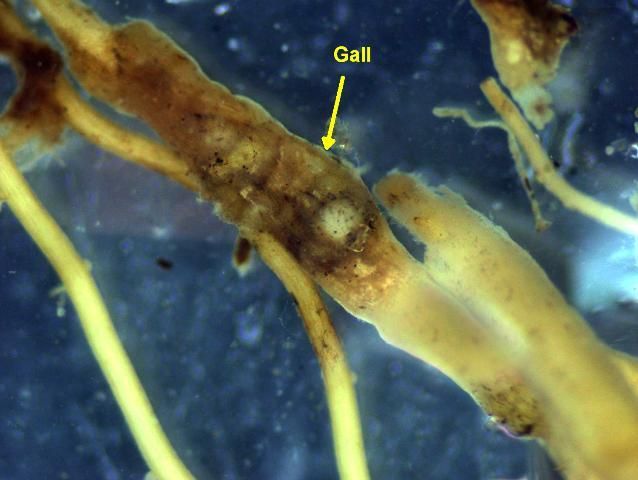
All of the plant-parasitic nematodes that damage turf in Florida feed on roots. As plant-parasitic nematodes feed, they damage plant root systems and reduce their ability to obtain water and nutrients from the soil. This makes the turf more susceptible to drought and other stresses. Nematode-damaged roots may be abnormally short and stubby (Figure 6) or appear darkened or rotten when damaged by plant-parasitic nematodes (Figure 7). Often the roots will appear "cropped off" an inch or less below the soil surface (Figure 8). Root galls or knots caused by certain species of nematodes to other crops are usually not evident on grasses but may occur in some cases (Figure 9).

Credit: W. T. Crow, UF/IFAS
Credit: R. A. Dunn, UF/IFAS

Credit: W. T. Crow, UF/IFAS
Credit: W. T. Crow, UF/IFAS
When nematode population densities get high enough, or when environmental stresses occur, above-ground symptoms may become evident. Symptoms include yellowing, wilting, browning, or thinning out. Grass will die under extreme nematode and environmental stress. Often, as the grass thins out spurge and other weeds may become prominent (Figure 10). Nematode damage usually appears as irregularly shaped declining areas that may enlarge slowly over time (Figure 11). Nematode symptoms on turfgrasses are often misdiagnosed because they appear similar to other factors such as localized soil conditions, fungal diseases, or insects.

Credit: W. T. Crow, UF/IFAS

Credit: W. T. Crow, UF/IFAS
Grass Species–Specific Associations
All types of grasses are affected by nematodes to some extent. However, certain nematodes are more problematic on some grasses than others.
Bahiagrass (Paspalum notatum) is the most tolerant turfgrass to plant-parasitic nematodes and rarely suffers damage from them. However, root-knot nematodes have recently been observed causing extensive damage to bahiagrass lawns.
St. Augustinegrass (Stenotaphrum secundatum) is the most common species of turfgrass used in Florida, and 'Floratam' is the most common cultivar of St. Augustinegrass. The most common nematodes affecting St. Augustinegrass are lance nematode, stubby-root nematode, and root-knot nematode. Floratam is relatively tolerant of sting nematode due to its thick root structure, but cultivars with finer roots are more susceptible.
Zoysiagrass (Zoysia spp.) is commonly damaged by sting, root-knot, stubby-root, and lance nematodes. No differences in nematode tolerance among zoysia cultivars have been identified.
Bermudagrass is commonly damaged by sting, root-knot, stubby-root, and lance nematodes. UF research found that among bermudagrass cultivars used in Florida, 'Celebration' and 'Princess 77' were more tolerant to sting nematode than 'Tifway' due to their vigorous rooting habit.
Centipedegrass (Eremochloa ophiuroides) is the most susceptible turfgrass to ring nematode and also is commonly damaged by sting, root-knot, stubby-root, and lance nematodes.
Seashore paspalum (Paspalum vaginatum) is fairly tolerant to most nematodes due to its vigorous root growth. However, it is very susceptible to spiral nematode. It also is occasionally damaged by sting and lance nematodes.
Nematode Diagnosis
With any plant problem, accurate diagnosis is important to address the problem and to avoid wasting time, money, effort, and pesticide applications. The only reliable way to determine if plant-parasitic nematodes are associated with grass damage is by having a nematode assay conducted by a professional nematode diagnostic lab. The University of Florida Nematode Assay Lab (NAL) is such a facility. Forms and instructions for submitting nematode samples to the Florida Nematode Assay Lab can be found on the NAL webpage at http://entnemdept.ufl.edu/nematology-assay-lab/.
A nematode assay requires different sampling guidelines than those required for soil analysis or disease samples. When a disease sample is submitted to most labs, a nematode analysis is not normally performed unless specifically requested. A nematode assay often requires separate payment and may even be sent to a separate address. Familiarize yourself with the procedures required by the lab where you intend to submit the sample. The accuracy of the diagnosis depends on the quality of the sample that you submit. Following the guidelines below will help ensure an accurate diagnosis:
- A sample must consist of multiple soil cores. Nematodes are not evenly distributed in soil, but their numbers may be high at one spot and low just a few feet way. By collecting multiple cores with a device such as a "T" type soil sample tube, an average population density can be measured. A good rule of thumb is to have 12–16 cores per lawn. Cores should be taken to a depth of 4 inches.
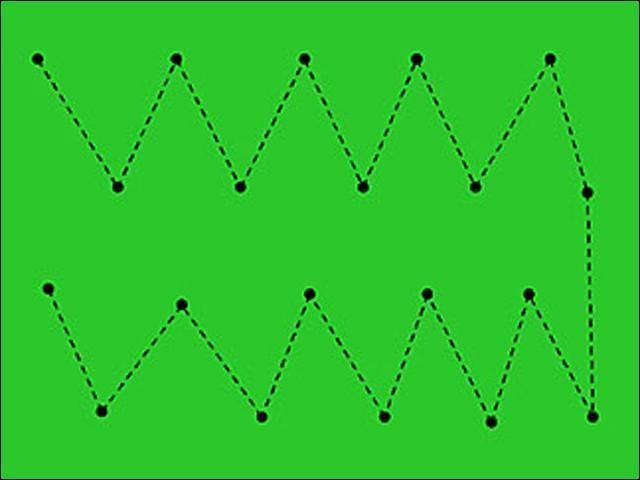
- If damage is evident, then sample near the margin of the affected area (Figure 12). Nematode populations will decline in severely damaged areas because they have nothing left to eat. Therefore, populations tend to be highest near the edges of a declining area where the grass is still alive. If damage is occurring in multiple areas of a field, take a few cores from the border of several affected areas to make the 12–16 cores. When taking samples from turf that is not showing symptoms, or if sampling before planting, sample in a "zig-zag" pattern across the area (Figure 13).
- Put the soil from each lawn into a separate plastic bag and seal it. Nematodes require moisture to survive, so drying the soil will kill them. This is different than submitting a sample for nutrient analysis where dry soil is preferred. Make sure that each bag is labeled with a permanent marker so that the diagnosis can be assigned to the correct area. If using a self-sealing bag, seal it with tape also because the zippers often come open in transit.
- Handle samples carefully. Do not expose samples to direct sunlight or heat. Nematodes are sensitive to high temperatures and UV light. Leaving samples on the dashboard or in the back of a pickup truck can kill them quickly and reduce the accuracy of the diagnosis. Keeping the nematode sample in a cooler is best. The nematodes will be sandwiched between soil particles so rough handling will destroy them. Pack the samples well to minimize shifting during shipping.
- Submit the sample right away. Next day delivery is best. One study found greatest nematode recovery from hand-delivered samples, the next highest from next-day delivery, and the lowest from regular postal delivery.
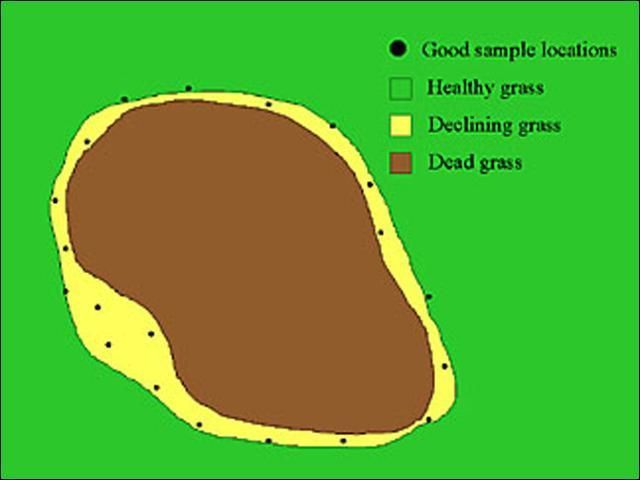
Credit: W. T. Crow, UF/IFAS
Credit: W. T. Crow, UF/IFAS
The NAL staff will identify any plant-parasitic nematodes found, count them, and determine if nematodes are a potential problem. Not all plant-parasitic nematodes are equal in their ability to damage grass. For example, one sting nematode can cause damage equal to hundreds of individuals of other types of plant-parasitic nematodes. The number of each type of nematode in 100 cc of soil from the submitted sample will be used to determine the risk level for the turf species indicated. The risk level will tell if the turf is at low, moderate, or high risk of damage from plant-parasitic nematodes. Be aware that different diagnostic labs may use different extraction techniques, different quantities of soil, or different thresholds. Because of this, samples submitted to separate labs may report different quantities of nematodes. In most cases the different thresholds used are adjusted to account for the differences in methodology and local conditions. However, if using a lab located in a geographically distant location, local conditions or regional variations in nematode aggressiveness may not be taken into account. Local labs will provide the most accurate assessments.
Nematode Management
Cultural Practices
Turf infested with plant-parasitic nematodes can often persist with no visible damage. Damage usually becomes evident when one of two things occur; 1) some other factor increases the susceptibility of the grass to nematode damage and/or 2) some factor causes nematode population densities to increase to damaging levels. Once the grass is planted, the best way to reduce the likelihood of nematode damage is to minimize these factors as much as possible. Therefore, some of the best practices for managing nematode damage in home lawns target avoiding other stresses on the grass. Properly irrigated and fertilized grass can often withstand higher levels of nematode infestation than grass suffering from drought or nutrient deficiencies. Following University of Florida Best Management Practices should ensure a properly cared-for lawn.
Research at the University of Florida has found that incorporation of certain organic amendments such as Comand® compost can improve tolerance to nematodes and in some cases suppress nematode numbers. Organic amendments such as Comand® compost, mulch, municipal sludge, and manure can help the grass tolerate nematode damage by improving plant health. Remember that anything that can be done to improve root health will help plants tolerate nematode damage. In addition, some organic amendments may stimulate soil microbes that can help suppress nematodes in some situations. Over-compaction reduces oxygen availability to the root system and enhances susceptibility to nematode damage. Regular aeration encourages a healthy root system and thereby enhances tolerance to nematodes. If areas are in partial shade, trimming or thinning trees to increase light to the turf will greatly enhance the turf's ability to withstand nematode damage. Too much shade may also mean that the site is not suitable for a dense stand of turfgrass. UF research has shown that overseeding can increase nematode numbers on bermudagrass during transition by providing an alternate food source to the nematodes during the winter. Therefore, it is best to avoid overseeding when possible, from a nematode management standpoint.
Pesticides and Biopesticides
Indemnify®
The active ingredient of Indemnify nematicide is fluopyram, which is also a fungicide. Indemnify is very effective against most of the nematodes that damage turfgrasses in Florida and is labeled for all turf use sites, including golf courses, athletic fields, sod farms, and lawns. Unlike other nematicides, it has very long residual activity and can provide nematode suppression for months after application. The maximum broadcast rate for Indemnify on residential lawns is 15.5 fluid ounces per acre per year. However, areas of 10,000 ft2 or less (per acre) may be "spot-treated" up to 4 times per year at 15.5 fluid ounces per acre. While Indemnify has provided excellent control of root-knot, sting, and other nematodes in University of Florida trials, it has not been shown effective against lance nematode.
Nimitz Pro G®: The active ingredient in Nimitz Pro G® is fluensulfone, a nematicide in its own chemical class. This formulation is a granular product applied to the turf surface using a spreader and then moved into the soil with irrigation. Fluensulfone has both contact and systemic activity, so it is effective against nematodes in soil and inside roots, including lance nematode. Nimitz Pro G is labeled for all warm season turf uses, including golf course, athletic field, and lawn. It is not labeled for use on cool-season grasses due to phytotoxicity. A maximum of 240 pounds per acre per year may be applied, but this can be broken up into multiple applications of lower doses. In University of Florida trials best results have been achieved with either 4 monthly applications of 60 pounds per acre or 3 monthly applications of 80 pounds per acre.
Other Products
The University of Florida provides recommendations based on research trial results for efficacy and consistency. The products mentioned above have each been evaluated extensively. However, there are a number of other products that can be used legally for nematode management in lawns. Many of these are biologically derived. Some have been evaluated and not proven effective in UF research trials. Other products either have not been evaluated or are still being evaluated. That some products are not included here does not mean a specific product is ineffective, just that we do not have enough research evidence to say that it is effective.
Summary
At present, the best management strategies for nematodes in the home lawn are reducing stress, promoting root vigor, and choosing tolerant grasses, each of which increases the turf's ability to tolerate nematode damage. Researchers at the University of Florida are continually looking at new management options and are committed to bringing you the most current management recommendations possible. Consequently, this document will be modified with each breaking development.
Additional Information
For additional information regarding nematodes, nematode management, or help interpreting nematode assay results contact:
Dr. W. T. (Billy) Crow, Professor of Nematology, Phone: (352) 273-3941, Email: wtcr@ufl.edu.
For information on submitting samples to the UF/IFAS Florida Nematode Assay Lab or to check on the status of a sample you submitted contact:
UF/IFAS Florida Nematode Assay Lab, Phone: (352) 392-1994, Email: nemalab@ifas.ufl.edu, website: http://entnemdept.ufl.edu/nematology-assay-lab/