Introduction
Cryptocaryon irritans (Cryptocaryon) is a ciliated, protozoan parasite which causes a disease known as marine "ich" or marine "white spot" disease in wild and cultured marine fishes at temperatures between 15 and 30°C (59–86°F) (Burgess and Matthews 1995; Diggles and Lester 1996a; Colorni and Burgess 1997). Cryptocaryon infections in fish are a significant disease problem for marine aquarists and commercial mariculture worldwide. Cryptocaryon is known to infect many different fish species, although there appear to be differences in susceptibility (Wilkie and Gordin 1969; Colorni 1985; Colorni and Burgess 1997).
Strain differences among different Cryptocaryon isolates from various parts of the world have been identified, and, although many have similar life cycles and salinity tolerances, others have been found to be outside previously described "normal" ranges (Yambot et al. 2003). The possibility of different species of Cryptocaryon also has been suggested; but all Cryptocaryon isolates currently are considered to be members of one species (Colorni and Burgess 1997; Yambot et al. 2003).
Signs of infection, complex life cycle stages, and the explosiveness with which infection and deaths can occur—often within days in culture situations—(Colorni and Burgess 1997) are similar in many ways to those seen for the freshwater parasite Ichthyophthirius multifiliis (Floyd and Reed 2009). However, the two are only distantly related, and major differences exist with regard to salinity tolerance and duration of life cycle.
Accurate diagnosis and rapid response and treatment are necessary to reduce losses, which can be devastating.
Signs of Disease
Fish infected with Cryptocaryon will often have small white spots, nodules, or patches on their fins, skin, or gills (Figure 1). They may also have ragged fins, cloudy eyes, pale gills, increased mucus production, or changes in skin color, and they may appear thin (Noga 1996; Colorni and Burgess 1997). Because the characteristic white spots might not be obvious in pale-colored fish or may not appear at all in infections with only gill involvement, absence of "white spots" or nodules—or even parasites—on the fin or skin does not rule out Cryptocaryon. Examine the gills to be sure. Behaviorally, fish may flash (scratch), swim abnormally, hang at the surface or on the bottom, act lethargic, or breathe more rapidly as if in distress (Colorni and Burgess 1997). Within a population, mortalities may increase rapidly over the course of several days. However, the extent of pathology will differ depending upon the strain of parasite, the species of fish, previous exposure to the parasite, and the temperature of the water.

Credit: E. J. Noga
Biology and Life Cycle of Cryptocaryon irritans
Cryptocaryon irritans has a direct life cycle (Colorni and Burgess 1997). This means that infection can spread within a group of fish without the need for another animal host for development. However, the life cycle is complex and includes stages that develop on and off the fish (Figure 2). Each stage will be described in more detail below, after this brief description. The trophont is the "feeding stage," during which the parasite is found embedded within the tissues of the fish. After the trophont leaves the fish, it becomes a protomont before encysting and transforming into a tomont, or "reproductive stage." The tomont develops and divides into numerous tomites, which eventually leave the cyst as theronts, the free-swimming infective stages. Theronts actively seek fish hosts.
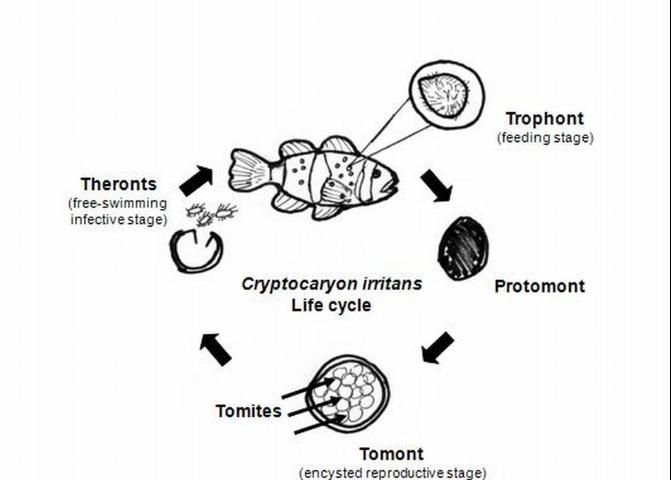
Credit: Roy Yanong, UF/IFAS
The length of the entire life cycle varies, depending upon a number of factors, including strain of Cryptocaryon, temperature, salinity, and fish host (Colorni 1985; Diggles and Lester 1996a, b, c; Colorni and Burgess 1997; Yambot et al. 2003). Even for a specific strain and fish host, the life cycle may vary by weeks or months (Colorni and Burgess 1997). An average life cycle appears to be 1 to 2 weeks; however, life cycle durations may range from 6 days to 11 weeks, primarily because of the unpredictability of tomont development (Colorni and Burgess 1997; Dickerson 2006; Yambot 2003).
In addition, some characteristics of the different life cycle stages (e.g., size and time required for development) vary depending upon the strain of Cryptocaryon irritans, the salinity and temperature of the water, and the species of fish infected (Diggles and Lester 1996 a, b, c).
Temperatures for optimal growth of most strains of Cryptocaryon appear to be about 23–30°C (73.4–86°F) (Dickerson 2006; Yoshinaga 2001), although active infections at 15°C (59°F) have been documented (Diggles and Lester 1996). Encysted stages, off the host (tomonts), were also observed to survive for 2–4 weeks under experimental hypoxic conditions (24% oxygen saturation); these released free-swimming infective stages (theronts) 10–11 days after excystment (Yoshinaga 2001).
A more recent study demonstrated that two life stages of one strain of Cryptocaryon (trophonts, i.e., the feeding stage during which the parasite can be found on the fish, and tomonts) survived dormant for 4–5 months at 12°C (53.6°F), and, after the water temperature increased to 27°C (80.6°F), developed and infected fish (Dan et al. 2009).
The most commonly observed stage is the trophont, or "feeding" stage. The trophont is found on the fish, usually underneath the outer skin layers. Spherical to club- or pear-shaped, with cilia all over its body, the trophont will normally be seen "rolling" or rotating slowly under the epithelium (outer cell layers of the skin or gills) (see Figures 3–6 ). Because the trophont is embedded within the skin, it is relatively protected from any potential treatments. The organism's cytoplasm is more opaque in this phase, which means the lobed macronucleus (and several smaller micronuclei) can be difficult to see in live specimens (Dickerson 2006). Trophonts can range in size from about 48 x 27 µm (~1/20 x 1/40 of a mm) to 452 x 360 µm (~1/2 x 1/3 mm) (1 µm = 1 micron; 1,000 microns = 1 millimeter). The trophont feeds on the body fluids and cells of the fish for about 3–7 days before leaving the host. Trophonts will also actively leave fish that have died, but are not immediately infective. They require additional time to develop from protomonts to tomonts, just as they would if leaving a live host (described below).

Credit: E. J. Noga
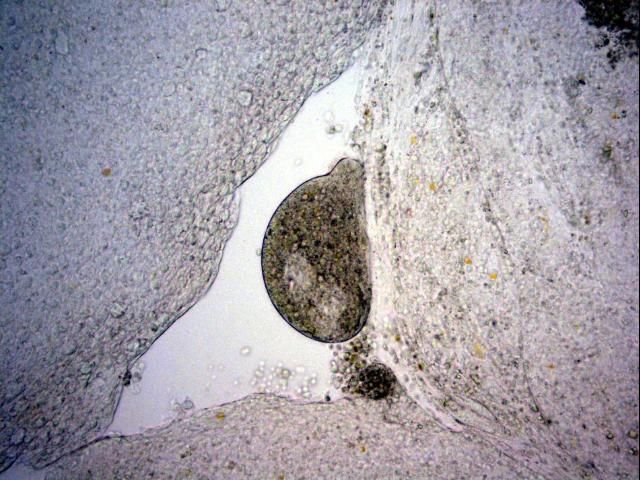
Credit: Georgia Aquarium, Veterinary Services Department
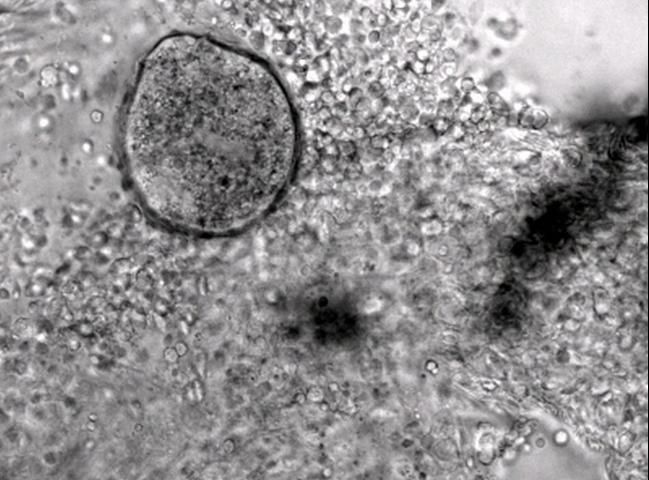
Credit: New England Aquarium
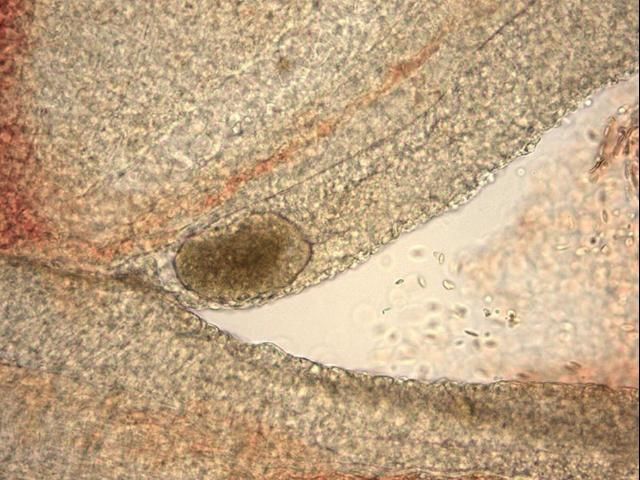
Credit: Georgia Aquarium, Veterinary Services Department
Once a trophont leaves the fish, it becomes a protomont. During this phase, it loses its cilia, flattens its surfaces, and moves onto a substrate for about 2–18 hours. After this stage, the organism stops, sticks to the surface, and encysts, whereupon it becomes a tomont. The cyst hardens in about 8–12 hours (Colorni 1985). Before the cyst forms, the protomont may be susceptible to some treatments for a short period of time. However, once the cyst has formed and hardened around the tomont, it has greater protection against common treatments
Tomonts range in size from 94.5 x 170 µm (~ 1/10 mm x 1/6 mm) to 252 x 441 µm (~1/4 x 1/2 mm). The tomont of one strain of Cryptocaryon was 210 x 763 μm (~1/5 x 3/4 mm). The encysted tomont undergoes many divisions, producing numerous daughter tomites (approximately 100 to 1000, depending upon strain and temperature [Colorni and Burgess 1997]). These tomites are released as theronts, the free-swimming infective stage.
The time required for theront development varies. In one study (Colorni and Burgess 1997), theronts emerged from a group of tomonts sometime between 3 and 72 days, with most released from 4 to 8 days after tomont formation. In another study (Diggles and Lester 1996c ), tomite development and theront release occurred, on average, between 5 and 12.1 days after tomont formation, depending upon strain and temperature. There was no correlation between tomont size and theront release. Yoshinaga and Dickerson (1994) observed, in laboratory studies, that theronts were released only between the hours of 2:00 am and 9:00 am, even in total darkness; some suggest this strategy increases the chance for theronts to find a host, as many fish may be resting or closer to substrate during this time period.
Theronts are oval to pear-shaped and motile; they actively seek fish. The theront is the most exposed, unprotected life stage and, therefore, the most logical target for treatment. Once the theront locates a host, it invades its skin within 5 minutes (Dickerson 2006). During gill invasion, the parasite becomes enclosed by a thin layer of cells within 20–30 minutes (Dickerson 2006). Theronts of one strain were 20–30 x 50–70 µm (Colorni 1985), but size will vary depending upon strain, host species, and temperature. The theront's infectivity is highest early in its life. By 6–8 hours after it leaves the cyst, its infectivity is greatly reduced (Burgess 1992; Yoshinaga and Dickerson 1994; Colorni and Burgess 1997; Dan et al. 2009), although a non-infective theront may still be able to move for up to 48 hours.
Immunity
As is seen with other diseases, general fish health and environmental factors including water quality will affect the status of the fish's immune system and may worsen the effects of an infection. If the immune status of the fish is compromised or if environmental factors are less than optimal, Cryptocaryon infection will be even more explosive and harmful.
Fish that survive a Cryptocaryon infection develop immunity, which can prevent significant disease for up to 6 months (Burgess 1992; Burgess and Matthews 1995). However, these survivors may act as carriers and provide a reservoir for future outbreaks (Colorni and Burgess 1997).
More targeted development of a vaccine to protect against Cryptocaryon irritans has been ongoing for a number of years (Yambot and Song 2006; Hatanaka 2007; Luo et al. 2007; Bai et al. 2008), and preliminary results are encouraging. However, vaccine development is a lengthy process, and no commercial vaccines are currently available.
Diagnostics
Cryptocaryon infection can be difficult to diagnose. Although white spots, nodules, or patches may be readily visible on the fish, other diseases can cause similar signs. Also, because of fish coloration or potential for infection only in the gills, these lesions may not be easily visible. Microscopic evaluation of skin, fin, and gill samples and identification of the trophont—spherical to pear-shaped, ciliated, opaque, rolling (see Figures 3–6)—or of one of the other life stages, is required to verify infection.
Cryptocaryon may be confused with other ciliates, including Uronema or even Brooklynella, by less experienced aquaculturists. Work with a fish health specialist to get a correct diagnosis and proper treatment and prevention recommendations.
Prevention and Control
An understanding of Cryptocaryon's life cycle provides a scientific framework for disease prevention and management. The ultimate goal of a prevention or control program is to break the life cycle of the parasite and stop future infections.
How long each life stage will need for development will depend upon the fish species affected, the fish's immune status, the strain of Cryptocaryon, and environmental factors including temperature and salinity. However, the wide variability and length of the Cryptocaryon life cycle, and, in particular, the time required for tomite development and theront release; the presence of protected, "embedded" and encysted stages on and off the fish; and the potentially devastating consequences of an outbreak of this infection necessitate a prolonged quarantine and treatment period. A minimum quarantine period of 3–6 weeks at 24–27°C (75.2–80.6°F), is advised, and longer time frames (e.g., 7–11 weeks) may be necessary.
The water in the affected system must be treated in some way to kill the theronts living in the water column. Likewise, substrate (including, possibly, parts of the fish) may harbor encysted tomonts, and, therefore, tanks and associated substrate and materials will act as "incubators" of Cryptocaryon. Clean or replace these to help to reduce reinfection.
Review local, state, and federal regulations to ensure legal use before you employ any drug or chemical, and work closely with a fish health specialist. Take into consideration proper dosage, potential toxicities to fish or invertebrates, and environmental management. Options for food and game fish are much more limited, and regulations are more strictly enforced.
Use of ultraviolet (UV) sterilization to kill theronts has been suggested, based on research involving Ichthyophthirius multifiliis (freshwater "ich"). The recommended UV dose for Ichthyophthirius theronts is 100,000 µWsec/cm2 (Hoffman 1974). However, UV doses required for Cryptocaryon irritans are anecdotal or extrapolated, and range from 280,000 µWsec/cm2 (industry numbers) to 800,000 µWsec/cm2 (Colorni and Burgess 1997).
Theronts must go through the UV sterilization unit in order to be exposed, so any theronts that are not exposed to UV radiation and remain in the tank or holding areas will be unaffected. Similarly, encysted tomonts within the tank or holding area will not be affected.
Ozonation is a highly effective method for disinfection of water, but is more complicated and may affect water quality, especially with regard to reaction products in salt water. There is limited information on doses required to kill theronts or other life stages (Colorni and Burgess 1997). Speak with a manufacturer or extension agent to determine the feasibility of ozone for your systems.
Although generally not feasible for large populations, large or complex systems, or weak or debilitated fish, one approach that has been suggested is transfer of affected fish into new, bare bottom tanks every 3 days (Colorni 1987). Tanks are cleaned, disinfected (see Disinfection below), and dried between moves. This approach reduces or prevents tomont development on the substrates, and subsequent reinfection. Depending upon temperature, fish may need to be moved 3–5 times.
Several chemical treatment options have been used most commonly and effectively against Cryptocaryon in marine aquaria and aquaculture systems. Most treatments target the free-swimming theront. Standard immersion (bath) treatments include copper, considered by many to be the most effective; hyposalinity (reducing salt concentration); and chloroquine. Formalin, though less commonly described in the literature to treat Cryptocaryon, has also been used with varying success.
(See Table 1. Chemical treatment options for control of Cryptocaryon irritans)
Copper, in the form of copper sulfate pentahydrate (CuSO4•5H20; the "blue" copper), is perhaps the most commonly used chemical against external protistan (protozoal) parasites of marine finfish. When used in marine systems, copper sulfate pentahydrate is considered 25.5% active ingredient (i.e., 25.5% is active, "free copper" (Cu2+).
The recommended dose is 0.15–0.20 mg/L free copper (Cu2+) (Noga 1996); however, this treatment concentration should be attained gradually, preferably over the course of 2–3 days, and careful measurement during dosing is necessary. This "breaking in" period will allow the fish time to increase their ability to reduce the toxic effect of copper (de Boeck et al. 2003). Having and using a good copper test kit is important, because copper levels can fluctuate during treatment. Free copper levels should be checked at least twice a day to ensure that copper levels are within the desired range. For more information on copper use, see "The Use of Copper for Treatment of Marine Aquarium Fish" at https://edis.ifas.ufl.edu/FA165.
Because of the prolonged life cycle of Cryptocaryon, affected systems should be treated for a minimum of 3–6 weeks (Noga 1996; C. Innis, pers. comm., T. Clauss, pers. comm.). As described above, in some reports, theronts were not released until 72 days after initial tomont formation, so some situations may require longer treatment time periods.
Chelated copper (copper that has been bound or "complexed" to other substances, such as citrate or EDTA, to increase its stability in water) has also been used, but safety and effectiveness are more variable than with copper sulfate pentahydrate (Noga 1996). If using a commercial product, refer to the manufacturer's directions and/or speak with a company representative for best results. You should also contact the company's technical department for questions regarding accurate testing for copper concentrations.
Hyposalinity refers to exposure of fish to a salt concentration that is lower than that in which they normally live (typical tropical marine systems range between 30–35 g/L (g/L=ppt)). Lower salinities are less easily tolerated by many common marine tropical species, which prefer a tighter range of salinity (stenohaline). Therefore, for many species, the lower the salinity, the shorter the time period the fish can tolerate. Freshwater or lower salinity dips (duration in minutes) or short or prolonged immersion baths (duration in hours or days) for tolerant fish species are commonly used to kill or reduce the numbers of external parasites on marine species.
However, Cryptocaryon has proven to be more challenging to treat using salinity changes. Because trophonts and tomonts are more protected, longer dips and baths will be required than for many other species of parasites. Exposure to freshwater for up to 18 hours did not seem to affect Cryptocaryon trophonts on the host (Colorni 1985). Prolonged exposure to 15–16 g/L salinity or less (Cheung et al. 1979; Colorni 1985) appeared to affect some life stages. Tomonts of one strain of Cryptocaryon were effectively killed after 48 hours of exposure to 15 g/L or less (Colorni 1985). Temperature will also determine whether hyposalinity will control the parasite, with temperatures outside the optimal range (23–30°C) causing greater breakdown of tomonts (Cheung et al. 1979).
More recently, studies have demonstrated different salinity tolerances among strains of Cryptocaryon. Yambot (2003) described one Taiwanese outbreak occurring in sea bream Sparus sarba at a salinity of 5 g/L, and another outbreak in sea perch Lates calcarifer occurring at a salinity of 10 g/L. These two strains were successfully propagated in the laboratory at 7 and 10 g/L, respectively, and are well below previously documented preferred salinities.
One suggested protocol that may have some effectiveness, depending upon temperature and the strain's salinity tolerance, is to maintain water at 15 g/L for 21–30 days (Noga 1996; Kinsler, pers. comm.). Salinity should be reduced gradually by 5 to 10 g/L per day until 15 g/L is reached.
Chloroquine, a quinine derivative, and other related compounds have been recommended for use against Cryptocaryon and other protozoan parasites including Amyloodinium (Dickerson 2006; Stoskopf 1993; Noga 1996; Roberts et al. 2009; I Berzins, pers. comm.; T. Clauss, pers. comm.). One recommended treatment regimen is 10 mg/L chloroquine diphosphate as a prolonged bath; duration of 2 to 3 weeks or more may be required. Chloroquine appears to be fairly stable. If water changes are necessary, redose in amounts proportional to quantity of water removed.
Formalin has been used with variable success and differing dosage regimens (Hoff 1996; Colorni and Burgess 1997; R. Francis-Floyd and D. Petty pers. comm. 2009). One suggested regimen is concurrent hyposalinity (16–18 g/L) and 25 mg/L formalin every other day, for 4 weeks (R. Francis-Floyd and D. Petty pers. comm.). Consider species sensitivities and specific system idiosyncrasies, as well as properties of formalin; be aware of these prior to use (Francis-Floyd 1996).
Disinfection
A number of methods for disinfection of Cryptocaryon tomonts and theronts have been examined (Hirazawa et al. 2003). The following regimens were shown to be effective against tomonts in the laboratory: 1 hour exposure to 40°C (104°F) or to 100 mg/L benzalkonium chloride; 24 hours exposure to 60 mg/L chlorine or to drying. Free-swimming theronts were killed more easily after one of the following treatments: 1 hour exposure to 2.4 mg/L chlorine; 1 hour exposure to 100 mg/L benzalkonium chloride; or 40°C (104°F).
(See Table 2. Methods for disinfection of Cryptocaryon irritans life stages)
Biosecurity Considerations
A good biosecurity program, developed with the assistance of a fish health specialist, will include quarantine of new fish (Yanong 2009). Quarantine marine plants, invertebrates (including shrimp, clams, corals, live rock), tank substrate, and other materials that may have been exposed to infected fish and/or harbor tomonts (reproductive, encysted stages) before introducing them into a system. Any used equipment or tanks should be properly disinfected prior to reuse. Aerosolization of theronts or other life stages from one tank to another is likely possible, especially if tanks are heavily aerated and air movement is favorable. Aerosolization as a method of spreading parasites has been demonstrated with Ichthyophthirius (Wooster et al. 2001). Source water (e.g., raw seawater) is a potential reservoir.
Ideally, you should sample skin, fins, and gills of fish for presence of Cryptocaryon before, during, and after quarantine (and treatment, if an infection is present) to determine if additional time is required. Use of hyposalinity, drugs, or chemicals during quarantine should also further reduce chance of spread. Trophonts on the fish and encysted stages (tomont) off the fish are well-protected against many common treatments. The free-swimming theronts are considered the most susceptible stage, and are the target of most treatments.
However, keep in mind that carriers may also be present in the established (resident) population and thereby infect any new, naïve fish that have been added to that system, even if these new fish were quarantined.
Summary
Cryptocaryon irritans, the causative agent of "marine white spot disease" is an important disease of marine and brackish water finfish, and has been documented in aquacultured, captive display, and wild populations. Common disease signs include white spots or areas of increased mucus, flashing, or respiratory distress; however, other signs may be seen, and fish should be sampled by a fish health specialist to verify presence of the parasite with a microscope.
A number of factors determine how severe the disease and mortalities will be and the length of the parasite life cycle. These factors include the strain of Cryptocaryon, the temperature and salinity of the water, the species and age of the fish and their general immune status, previous exposure to the parasite, the number of infective parasites present, and the dissolved oxygen concentration of the water. The life cycle can last from 6–11 weeks, but an average parasite life cycle appears to be 1–2 weeks. However, because the time required until release of infective theronts after tomont formation varies so widely, prolonged treatment periods (3–6 weeks) are recommended. Longer treatment time may be necessary.
Treatments recommended in this publication for any given outbreak should provide significant mitigation of disease. Copper sulfate pentahydrate appears to be the most effective treatment to date. Chloroquine and salinity, as well as formalin, have also proven to have some effectiveness. If the Cryptocaryon strain affecting your fish is tolerant of low salinity, however, hyposalinity will not be an effective control.
Quarantine of any new fish for 30–90 days before introducing them into an established population will provide time for observation, treatment, and reduction of spread. Source water should also be considered a potential reservoir, and, therefore, should be handled appropriately. In addition, any equipment, tank structures, or other inert materials should be disinfected properly prior to reuse in other systems. Clinically healthy fish that have survived an infection may act as carriers. Similarly, slow developing or dormant tomonts in the environment may act as a reservoir.
Working with a fish health specialist is highly recommended to help design biosecurity/quarantine and treatment regimens.
Acknowledgements
Thanks to Charlie Innis and Sarah McCormack, Tonya Clauss, Andy Stamper, James Kinsler, Ilze Berzins, Edward Noga, Ruth Francis-Floyd, and B. Denise Petty for assistance with this publication.
References and Suggested Reading
Bai, J.S., M.Q. Xie, X.Q. Zhu, X.M. Dan, and A.X. Li. 2008. Comparative studies on immunogenicity of theronts, tomonts and trophonts of Cryptocaryon irritan in grouper. Parasitol Res 102:307–313.
Burgess, P.J. 1992. Cryptocaryon irritans Brown, 1951 (Ciliophora): transmission and immune response in the mullet Chelon labrosus (Risso, 1826). Ph.D. thesis, University of Plymouth.
Burgess, P.J. and R.A. Matthews. 1995. Cryptocaryon irritans (Ciliophora): acquired protective immunity in the thick-lipped mullet, Chelon labrosus. Fish and Shellfish Immunology 5: 459–468.
Cheung, P.J., R.F. Nigrelli, and G.D. Ruggieri. 1979. Studies on cryptocaryoniasis in marine fish: effect of temperature and salinity on the reproductive cycle of Cryptocaryon irritans Brown, 1951. Journal of Fish Diseases 2: 93–97.
Colorni, A. 1985. Aspects of the biology of Cryptocaryon irritans, and hyposalinity as a control measure in cultured gilt-head sea bream Sparus aurata. Diseases of Aquatic Organisms 1: 19–22.
Colorni, A. 1987. Biology of Cryptocaryon irritans and strategies for its control Aquaculture 67: 236–237.
Colorni, A. and P. Burgess. 1997. Cryptocaryon irritans Brown 1951, the cause of "white spot disease" in marine fish: an update. Aquarium Sciences and Conservation, 1: 217–238.
Dan, X.M., A.X. Li, X.T.Lin, N.Teng, and X.Q. Zhu. 2006. A standardized method to propagate Cryptocaryon irritans on a susceptible host pompano Trachinotus ovatus. Aquaculture 258: 127–133.
Dan, X.M., X.T. Lin, Y.X Yan, N. Teng, Z.L. Tan, and A.X. Li. 2009. A technique for the preservation of Cryptocaryon irritans at low temperatures. Aquaculture 297: 112–115.
De Boeck, G., T.T.H. Ngo, K. Van Campenhout, and R. Blust. 2003. Differential metallothionein induction patterns in three freshwater fish during sublethal copper exposure. Aquatic Toxicology 65; 413–424.
Dickerson, H.W. 2006. Ichthyophthirius multifiliis and Cryptocaryon irritans (Phylum Ciliophora). Pages 116–153 In Woo, P.T.K., ed. Fish diseases and disorders vol.1: protozoan and metazoan disorders. 2nd ed. CAB International. Cambridge, MA.
Diggles, B.K. and R.D. Adlard. 1997. Intraspecific variation in Cyrptocaryon irritans. J. Euk. Microbiol. 44 (1): 25–32.
Diggles, B.K. and R.J.G. Lester. 1996a. Infections of Cryptocaryon irritans on wild fish from southeast Queensland, Australia. Diseases of Aquatic Organisms 25: 159–167.
Diggles, B.K. and R.J.G. Lester. 1996b. Influence of Temperature and Host Species on the development of Cryptocaryon irritans. The Journal of Parasitology Vol. 82(1): 45–51.
Diggles, B.K. and R.J.G. Lester. 1996c. Variation in the development of two isolates of Cryptocaryon irritans. The Journal of Parasitology Vol. 82 (3): 384–388.
Francis-Floyd, R. 1996. Use of formalin to control fish parasites. VM-77. College of Veterinary Medicine, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL. https://edis.ifas.ufl.edu/VM061 (Accessed November 12, 2009)
Francis-Floyd, R. and P. Reed. 2009. Ichthyophthirius multifiliis (white spot) infections in fish. CIR920. Program in Fisheries and Aquatic Sciences, SFRC, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL. https://edis.ifas.ufl.edu/FA006 (accessed November 12, 2009)
Hatanaka, A., N. Umeda, S. Yamashita, and N. Hirazawa. 2007. Identification and characterization of a putative agglutination/immobilization antigen on the surface of Cryptocaryon irritans. Parasitology 134: 1163–1174.
Hirazawa, N., T. Goto, and K. Shirasu. 2003. Killing effect of various treatments on the monogenean Heterobothrium okamotoi eggs and oncomiracidia and the ciliate Cryptocaryon irritans cysts and theronts. Aquaculture 223: 1–13.
Hoff, F.H. 1996. Conditioning, spawning, and rearing of fish with emphasis on marine clownfish. Aquaculture Consultants, Inc., Dade City, FL. 212 pp.
Hoffman, G.L. 1974. Disinfection of contaminated water by ultraviolet irradiation, with emphasis on whirling disease (Mysoxosma cerebralis): and its effect on fish. Transactions of the American Fisheries Society 103: 541–550.
Luo, X.C., M.Q. Xie, X.Q. Zhu, and A.X. Li. 2008. Some characteristics of host-parasite relationship for Cryptocaryon irritans isolated from South China. Parasitology Research 102: 1269–1275.
Noga, E.J. 1996. Fish disease: diagnosis and treatment. Mosby-Year Book, Inc., St. Louis, MO. 367 pp.
Roberts, H., B. Palmiero, and E.S.Weber III. 2009. In Bacterial and parasitic diseases of fish. Laura Wade, ed. Veterinary clinics of North America: exotic animal practice. 12 (3): 609–638.
Stoskopf, M.K. 1993. Fish medicine. W.B. Saunders, Philadelphia, PA. 992 pp.
Wilkie, D.W. and H. Gordin. 1969. Outbreak of cryptocaryoniasis in marine aquaria at Scripps Institution of Oceanography. California Fish and Game 55(3): 227–236.
Wooster, G.A., T.M. Bishop, and P.R. Bowser. 2001. The aerobiological dissemination of the fish parasite Ichthyophthirius multifiliis. Proceedings, 26th Eastern Fish Health Workshop. Leetown, WV.
Yambot, A.V., and Y.L. Song. 2006. Immunization of grouper, Epinephelus coioides, confers protection against a protozoan parasite, Cryptocaryon irritans. Aquaculture 260: 1–9.
Yambot, A.V., Y.L. Song, and H.H. Sung. 2003. Characterization of Cryptocaryon irritans, a parasite isolated from marine fishes in Taiwan. Diseases of Aquatic Organisms 54: 147–156.
Yanong, R.P.E. 2009. Fish health management considerations in recirculating aquaculture systems. Program in Fisheries and Aquatic Sciences, SFRC, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL. https://edis.ifas.ufl.edu/FA100 (accessed November 12, 2009)
Yanong, R.P.E. 2009 (in review). Use of copper in marine aquaculture and aquarium systems. Program in Fisheries and Aquatic Sciences, SFRC, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, FL. https://edis.ifas.ufl.edu/FA165
Yoshinaga, T. 2001. Effects of high temperature and dissolved oxygen concentration on the development of Cryptocaryon irritans (Ciliophora) with a comment on the autumn outbreaks of Cryptocaryoniasis. Fish Pathology 36(4): 231–235.
Yoshinaga, T. and H.W. Dickerson. 1994. Laboratory propagation of Cryptocaryon irritans on a saltwater-adapted Poecilia hybrid, the Black Molly. Journal of Aquatic Animal Health 6: 197–201.