The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Introduction
The citrus leafminer (CLM), Phyllocnistis citrella Stainton, is a potentially serious pest of citrus and related Rutaceae and some related ornamental plants (Beattie 1989, Clausen 1933, Kalshoven 1981). CLM (Figure 1) was previously intercepted in the United States in 1914 (ports not noted) on citrus and Atalantia horticulture stock imports from the Philippines (Sasscer 1915). In 1993, when it was finally discovered in Florida, it was a new record for Florida, the continental United States, and the Western Hemisphere.

Credit: Lyle J. Buss, UF/IFAS.
Distribution
A widespread Asian species (Clausen 1931, 1933, CAB 1970), described from Kolkata, India (Stainton 1856), CLM is known from East Africa—Sudan to Yemen (Badawy 1967) and through southern Asia—Saudi Arabia to India (Fletcher 1920) and Indonesia (Kalshoven 1981), north to Hong Kong and China, the Philippines (Sasscer 1915), Taiwan (Chiu 1985, Lo and Chiu 1988), and southern Japan (Clausen 1927). It is also found in New Guinea and nearby Pacific Islands (CAB 1970). It was introduced into Australia before 1940, and by 1995 had spread across the continent (Beattie and Hardy 2004). CLM also occurs in South Africa and in parts of West Africa (CAB, personal communication).
In the Western Hemisphere, CLM was first discovered in Florida in May 1993 in several citrus nurseries in Homestead, other parts of Miami-Dade County, and in Broward and Collier counties. Since then, it has spread to all Florida citrus counties. Since 1994, it has spread to Alabama, Louisiana, and Texas (Nagamine and Heu 2003). By 1995, the citrus leafminer was discovered in Central America, western Mexico, and several Caribbean islands (Jones 2001). In 2000, it arrived in southern California from Mexico (Grafton-Cardwell et al. 2019), and was first detected in Hawaii on Oahu, spreading to Kauai and Maui in 2001 and Molokai and Hawaii (the Big Island) in 2002 (Nagamine and Heu 2003).
Description
Adults of the CLM (Figure 2) are minute moths with a 4 mm wingspread. They have white and silvery iridescent scales on the forewings, with several black and tan markings, plus a black spot on each wingtip. The hind wings and body are white, with long fringe scales extending from the hindwing margins. In resting pose with wings folded, the moth is much smaller in appearance (about 2.4 mm). The head is very smooth-scaled and white, and the haustellum has no basal scales. CLM is most easily detected by its meandering serpentine larval mine, usually on the ventral side of the leaf. Larvae are minute (up to 3 mm), translucent greenish-yellow, and located inside the leaf mine. The pupa characteristically is in a pupal cell at the leaf margin. Adults generally are too minute to be easily noticed and are active diurnally and in the evenings.

Credit: Jeffrey W. Lotz, Florida Department of Agriculture and Consumer Services, Division of Plant Industry
Biology
The biology of CLM has been reported by a number of researchers, including Badawy (1967), Beattie (1989), Clausen (1927, 1931, 1933), Fletcher (1920), Kalshoven (1981), and Latif and Yunus (1951). Eggs of CLM are laid singly on the underside of host leaves. Egg eclosion occurs within two to 10 days, whereupon larvae immediately enter the leaf and begin feeding. Larvae make serpentine mines on young leaves (sometimes also young shoots), resulting in leaf curling and serious injury. Leaf mines are usually on the ventral leaf surface, except in heavy infestations when both leaf surfaces are used. Usually only one leaf mine is present per leaf but heavy infestations may have two or three mines per leaf, and up to nine mines on large leaves have been found in Florida.
As with similar leafminers, larvae are protected within the leaf during their feeding cycle. Larvae have four instars and development takes from five to 20 days. Pupation is within the mine in a special pupal cell at the leaf margin, under a slight curl of the leaf. Pupal development takes six to 22 days. Adults emerge about dawn and are active in the morning; other activity occurs at dusk or night. Females lay eggs evenings and at night (Badawy 1967, Beattie 1989). CLM may help spread citrus canker (Hill 1918; Ando et al. 1985) because of leaf damage from the mine.
Generations per year appear to be nearly continuous: six in southern Japan (Clausen 1931), nine to 13 in north-central India (Lal 1950), and 10 in southern India (Pandey and Pandey 1964). Development time totals about 13 to 52 days (Pandey and Pandey 1964). Depending on foliage flushing cycles and weather conditions, as many as six to 13 can be expected (Jones 2001). Adults live for only a few days. In Florida, a new generation is produced about every three weeks.
Host Plants
CLM is common on species of citrus and related Rutaceae within its range (Kalshoven 1981).
CLM is most commonly found on leaves of all citrus, including orange, lemon, lime, and tangerine.
Other Rutaceae recorded as hosts include:
- Aegle marmelos (L.) Corr. Serv. in India (Fletcher 1920),
- Atalantia sp. in the Philippines (Sasscer 1915),
- Murraya paniculata (L.) Jack. in India (Pruthi and Mani 1945),
- Poncirus trifoliata (L.) Raf. in India (Clausen 1933), and
- various native Rutaceae in Indonesia (Kalshoven 1981).
Other reported hosts include:
- Jasminum sambac (L.) Aiton (Oleaceae) in India (Fletcher 1920),
- mistletoes on citrus (Loranthus sp., Loranthaceae) in the Philippines (Reinking and Groff 1921),
- Pongamia pinnata Pierre (Leguminosae) in India (Margabandhu 1933), and
- Alseodaphne semecarpifolia Nees (Lauraceae) in India (Latif and Yunus 1951).
Several other hosts have been reported for CLM, but larvae do not complete their life cycle on these incompatible hosts:
- Murraya koenigii L. Sprengel (Rutaceae) in India (Fletcher 1920),
- Jasminum sp. and Jasminum cinnamomum Kobuski (Oleaceae) in India (Pruthi and Mani 1945),
- Dalbergia sissoo Roxb. ex DC (Leguminosae) in India (Latif and Yunus 1951),
- Salix sp. (Salicaceae) in India (Pruthi and Mani 1945), and
- Grewia asiatica L. (Tiliaceae) in India (Latif and Yunus 1951).
Damage
The larvae form serpentine mines in the leaves (Figure 3) and rarely fruit of their hosts. These mines are filled with a central line of frass. This characteristic helps separate this leafminer from the citrus peelminer. The citrus leafminer larvae only infest the younger, flushing foliage. Adults lay their eggs on both the upper and lower surface of leaves that are less than 1.27 cm (1/2 inch) in length. Reports from Australia indicate infestations of one to three mines per leaf, while wetter conditions in other areas, such as Florida, may support more miners per leaf (Jones 2001).
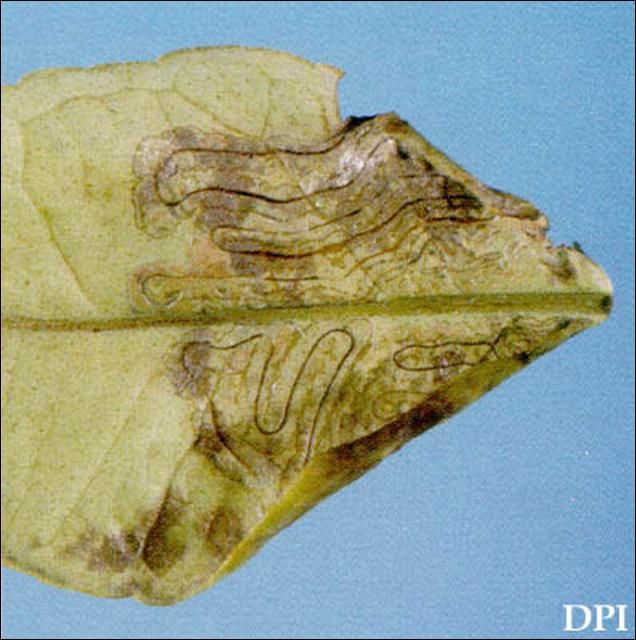
Credit: Jeffery W. Lotz, Florida Department of Agriculture and Consumer Services, Division of Plant Industry
Economic losses are reflected by the increased cost of protecting nursery and non-bearing citrus, as well as limiting sales of hosts to door-yard growers. Infestations can retard the growth of young trees (Grafton-Cardwell et al. 2019). Heavy infestations may affect production on mature trees, but this has been demonstrated only on limes (Jones 2001).
In Florida, a real concern is that the citrus leafminer creates openings that permit citrus canker bacteria to infest the tree (Grafton-Cardwell et al. 2019).
Survey
Symptoms of infestation include:
- leaves with serpentine mines, usually on ventral surfaces;
- curling of leaves (may harbor mealybugs);
- epidermis appearing as a silvery film over leaf mines;
- pupation chamber near leaf margin, the edge of which is rolled over, and exposed portion of chamber with a distinct orange color; and
- possibility of succulent branches of green shoots attacked (Beattie 1989, Pandey and Pandey 1964).
Management
Parasites reported for CLM include 39 species from Southeast Asia, Japan, and Australia, mostly Chalcidoidea (Heppner 1993, Kalshover 1981, Lo and Chiu 1988). A pheromone to attract males of CLM was developed in Japan by Ando et al. (1985), called (7Z, 11Z)-7, 11-hexadecadienal. Traps containing the pheromone are available and should be used to determine when the moths are flying (Grafton-Cardwell et al. 2019) Much work has been done using chemical control, especially in India. Various spray regimes, time of growth flushes, and promotion of biological control are recommended in Australia (Beattie 1989).
In Florida, biological control and applications of oil are suitable methods to help reduce populations of CLM. Natural enemies already present in Florida have responded to leafminer infestations, causing up to 90% mortality of larvae and pupae. These natural enemies include the introduced parasitoid Ageniaspis citricola that is established throughout most of Florida and is responsible for up to 30% of this mortality mostly later in the year (Rogers, Stansly, and Stelinski 2009). Apparently, Ageniaspis citricola was introduced into Hawaii at the same time as the citrus leafminer. Since then, Ageniaspis citricola has spread to all the islands and provides good control (Nagamine and Heu 2003).
As a result, treating with insecticides may affect parasites' populations and growers should consult with state experts on the timing and frequency of such treatments (Grafton-Cardwell et al. 2019).
For more on management, visit the Florida Citrus Pest Management Guide (https://edis.ifas.ufl.edu/publication/CG098).
Selected References
Ando T, Taguchi KY, Uchiyama M, Ujiye T, Kuroko H. 1985. (7Z-11Z)-7, 11-hexadecadienal sex attractant of the citrus leafminer moth, Phyllocnistis citrella Stainton (Lepidoptera, Phyllocnistidae). Agricultural and Biological Chemistry, Tokyo 49: 3633-3653.
Badawy A. 1967. The morphology and biology of Phyllocnistis citrella Staint., a citrus leaf miner in the Sudan. Bulletin of the Entomological Society of Egypt 51: 95-103.
Beattie GAC. 1989. Citrus leaf miner. NSW Agricultura & Fisheries, Agfact, H2. AE: 41-4.
Beattie A, Hardy S. (2004). Citrus leafminer. Department of Primary Industries, Industry & Investment New South Wales. (26 April 2013)
Chiu SC. 1985. Biological control of citrus pests in Taiwan. Taiwan Agricultural Research Institute, Special Report 19: 1-8.
Clausen CP. 1927. The citrus insects of Japan. USDA, Washington, D.C. Technical Bulletin 15: 1-15.
Clausen SP. 1931. Two citrus leaf miners of the Far East. USDA, Washington, D.C. Technical Bulletin 252: 1-13.
Clausen CP. 1933. The citrus insects of tropical Asia. USDA, Washington, D.C. Circular 266: 1-35.
Commonwealth Agricultural Bureaux (CAB), Commonwealth Institute of Entomology. 1970. Phyllocnistis citrella Stnt. In Distribution maps of pests. Ser. A, Map No. 274. The Eastern Press Ltd., London, England.
Fletcher TB. 1920. Life histories of Indian insects. Microlepidoptera. Memorandum of the Department of Agriculture, India 6: 1-217.
Grafton-Cardwell EE, Morse JG, O'Connell NV, Phillips PA, Kallsen CE, Haviland DR. (September 2009). Citrus leafminer. University of California IPM Online. (26 April 2010)
Grafton-Cardwell EE, Montez G. (April 2009). Citrus leafminer, Phyllocnistis citrella Stainton (Lepidoptera: Gracillariidae). Citrus Entomology, University of California. (26 April 2013)
Heppner JB. 1993. Citrus leafminer, Phyllocnistis citrella, in Florida (Lepidoptera Gracillariidae Phyllecnistinae). Tropical Lepidoptera 4: 49-64.
Hill GF. 1918. History of citrus canker in the Northern Territory (with notes of its occurrence elsewhere). Northern Territory Australia Bulletin 18: 1-8.
Jones J. (2001). Citrus leafminer. Arizona Crop Information Site. (26 April 2013)
Kalshoven LGE. 1981. Pests of crops in Indonesia. Jakarta: Ichtiar Baru. [reprint].
Lal KB. 1950. Insect-pests of fruit trees grown in the plains. Agric. Anim. Husb. Uttar Pradesh 1: 30-45.
Latif A, Yunus CM. 1951. Food plants of citrus leaf miner in Punjab. Bulletin of Entomological Research 42: 311-316.
Legaspi JC, French JV, Bendixen R. (December 1997). The citrus leafminer and its natural enemies. Texas A&M Agrilife. (26 April 2013)
Lo KC, Chiu SC. 1988. The illustrations of citrus insect pests and their natural enemies in Taiwan. Taichung Taiwan Agricultural Research Institute. 75 pp.
Margobandhu V. 1933. Insect pest of oranges in the northern Circars. Madras Agricultural Journal 21: 60-68.
Nagamine WT, Heu RA. (September 2003). Citrus leafminer, Phyllocnistis citrella Stainton. (26 April 2013)
Pandey ND, Pandey YD. 1964. Bionomics of Phyllocnistis citrella Stt. (Lepidoptera: Gracillariidae). Indian Journal of Entomology 26: 417-423.
Pruthi HS, Mani MS. 1945. Our knowledge of the insect and mine pests of the citrus in India and their control. Imperial Council Agricultural Research Sciences Monograph 16: 1-42.
Reinking OA, Groff GW. 1921. The kao pan seedless Siamese pummelo and its culture. Philippine Journal of Science 19: 389-437.
Rogers ME, Stansly PA, Stelinski LL. (November 2009). 2010 Florida Citrus Pest Management Guide: Asian citrus psyllid and citrus leafminer. EDIS. (26 April 2013)
Sasscer ER. 1915. Important insect pests collected on imported nursery stock in 1914. Journal of Economic Entomology 8: 268-270.
Stainton HT. 1856. Descriptions of three species of Indian Micro-Lepidoptera. Transactions of the Entomological Society of London (n.s.) 3: 301-304.
Stelinski LL, Miller JR, Rogers ME. 2008. Mating disruption of citrus leafminer mediated by a non-competitive mechanism at a remarkably low pheromone release rate. Journal of Chemical Ecology 34: 1107-1113.
Wilson CG. 1991. Notes on Phyllocnistis citrella Stainton (Lepidoptera: Phyllocnistidae) attacking four citrus varieties in Darwin. Journal of Australian Entomological Society 30: 77-78.