"Candidatus Liberibacter solanacearum": An Emerging Pathogen Infecting Potato and Tomato
Introduction
Florida is the second largest producer of tomatoes and the seventh largest producer of potatoes in the US. Among the major diseases of these vegetable crops, a new bacterial species "Candidatus Liberibacter solanacearum" has been found to infect potatoes, tomatoes, and other solanaceous crops in the US. The bacterium causes a destructive disease known as zebra chip in potatoes and psyllid yellows in tomatoes that results in significant economic loss by reducing both yield and quality of the crops. Yield losses of greater that 85% have been reported in potatoes and tomatoes (Trumble 2009). "Candidatus Liberibacter" species are obligate bacterial pathogens that inhabit the phloem cells of infected host plant species and are transmitted by the tomato/potato psyllid Bactericera cockerelli. This disease has not yet been detected in Florida.
Diseases associated with Ca. L. solanacearum include zebra chip in potatoes (Abad et al. 2009; Hansen et al. 2008; Leifting et al. 2008a; Leifting et al. 2009; Munyaneza 2012; Rehman et al. 2010; Secor et al. 2009) and psyllid yellows in tomatoes and peppers (Leifting et al. 2008a; Leifting et al. 2009; Munyaneza et al. 2009b). Ca. L. solanacearum has been reported to cause dieback and leaf curling in tamarillo (Solanum betaceum) (Leifting et al. 2008b; Watson 2009) and produces secondary root proliferation in carrots (Munyaneza et al. 2010). "Ca. L. solanacearum" has also been detected in several solanaceous weeds including Solanum ptycanthum (black nightshade), Solanum elaeagnifolium (silverleaf nightshade) and Lycium barbarum (wolfberry) and produces typical psyllid yellows symptoms (Wen et al. 2009).
Biology
Ca. L. solanacearum bacteria are Gram-negative, phloem-limited, unculturable bacteria belonging to the Alphaproteobacteria group. The bacterium has a rod-shaped morphology and is about 0.2 µm wide and 4 µm long. The bacteria closely resemble the Liberibacters associated with Huanglongbing (citrus greening) disease, but are heat-sensitive to temperatures above 32°C. Temperatures at or below 17°C may significantly slow the infection in potato (Munyaneza 2012; Munyaneza et al. 2012).
Distribution
"Ca. L. solanacearum" has been reported to occur in the western and central regions of the US, including Texas, Nebraska, Colorado, Kansas, Wyoming, New Mexico, Arizona, Nevada, California, Idaho, Oregon, and Washington (Abad et al. 2009; Brown et al. 2010; Crosslin and Bester 2009; Crosslin and Munyaneza 2009; Crosslin et al. 2012; Crosslin, Olsen, and Nolte 2012; Halbert and Munyaneza 2012; Hansen et al. 2008; McKenzie and Shatters 2009; Secor et al. 2009). The pathogen has also been reported in Mexico, Central America (Guatemala, Honduras, and Nicaragua), and New Zealand (Leifting et al. 2008a,b; Leifting et al. 2009; Munyaneza, Buchman, and Crosslin 2009; Munyaneza et al. 2009b; Rehman et al. 2010). Occurence of this species of Liberibacter on carrot (Daucus carota L.) has also been reported in Northern Europe and the Mediterranean Region (Spain and the Canary Islands) (Alfaro-Fernández et al. 2012a, b).
Symptoms
Symptoms associated with Ca. L. solanacearum in potato (Australian Department of Agriculture 2011):
- Yellowing or purpling of leaves and shoots (Figures 1 and 2)
- Leaf curling (Figures 1 and 2)
- Stunted shoots with shortened and swollen internodes (Figure 2)
- Aerial tuber formation (Figure 3)
- Appearance of scorched potato tops that collapse prematurely (Figure 4)
- Early senescence of the infected plants
- Tubers may have enlarged lenticels, with necrotic flecking of the vascular tissue and streaks along the medullary rays that are enhanced when slices of the potatoes are fried, and brown purplish stolons (Figure 5)

Credit: Dr. Lia Liefting

Credit: Dr. Lia Liefting

Credit: Dr. Lia Liefting

Credit: Dr. Lia Liefting
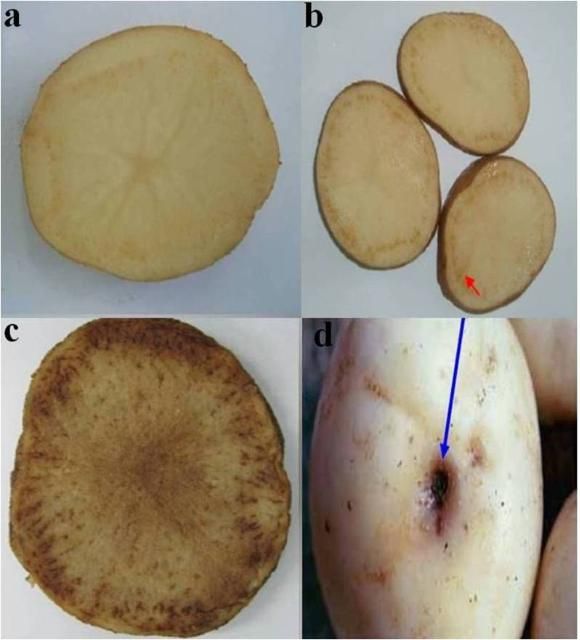
Credit: Dr. Lia Liefting
Symptoms associated with Ca. L. solanacearum in tomato (Australian Department of Agriculture 2011)
- Stunted growth (Figure 6)
- Spiky and chlorotic apical growth (Figures 6 and 7)
- Distorted and curled leaflets (Figures 6, 7, and 9)
- Mottled or chlorotic leaves (Figure 9)
- Interveinal chlorosis and vein greening of the leaves occurs in some varieties (Figure 6)
- Purpling of the midvein and leaves in some cultivars (Figure 10)
- Fruit deformation (Figure 8)

Credit: Dr. Lia Liefting

Credit: Dr. Lia Liefting

Credit: Dr. Lia Liefting

Credit: Dr. Lia Liefting

Credit: Dr. Lia Liefting
Mode of Transmission
Ca. L. solanacearum spreads mainly from plant to plant by grafting (Crosslin and Munyaneza 2009; Secor et al. 2009) and also by the psyllid vector B. cockerelli (Alfaro-Fernández et al. 2012b; Buchman, Sengoda, and Munyaneza 2011; Hansen et al. 2008; Munyaneza, Crosslin, and Upton 2007; Munyaneza et al. 2010; Secor et al. 2009). Ca. L. solanacearum is not transmitted through true seed from infected solanaceous plants.
Vector
The potato/tomato psyllid, Bactericera cockerelli (Sulc) (Hemiptera: Triozidae) is considered to be the causal vector of the Ca. L. solanacearum that causes zebra chip and psyllid yellows diseases. This psyllid feeds upon the phloem using piercing-sucking mouth parts.
Biology of the Vector
The adult potato psyllid is small in size, measuring 2.5 mm long (Figure 11 a and b), and has a body color of pale green at birth, dark green or brown within 2 to 3 days after birth, and eventually gray or black thereafter. These psyllids possess prominent white or yellow lines on the head and thorax. A distinguishing feature of B. cockerelli is the presence of a broad, white transverse band on the first abdominal segment and the inverted V-shaped white mark on the last abdominal segment (Wallis 1955). The adult psyllids are good fliers and may jump readily when disturbed.
The adult psyllids have a longevity ranging from 20 to 62 days, and females usually live two to three times longer than males (Abdullah 2008; Yang et al. 2010). The female lays an average of 300 to 500 light to dark yellow or orange eggs over her lifetime. The eggs are deposited singly and mounted on short stalks on the lower surface and edge of the leaf (Figure 11 c). The optimum development of the psyllids occurs at 27°C. Temperatures above 35°C result in a reduction of oviposition, hatching, and survival.
The nymphs of the psyllids are usually found on the lower surface of the leaves (Figure 11 d), and are orange or yellowish-green in color, and possess a prominent reddish eye. Potato psyllid nymphs may be easily confused with the nymphs of whiteflies. However, compared to whitefly numphs, potato psyllid nymphs moves only when disturbed.

Credit: Mike Lewis (a), Joseph E. Munyaneza (b) and J. Kelly Clark (c, d)
Vector Distribution
The potato psyllid is native to North America and is present in Arizona, Colorado, Idaho, Kansas, Nebraska, Nevada, New Mexico, North Dakota, Minnesota, Montana, Oklahoma, South Dakota,Texas, Utah, Wyoming, and as far west as California. The potato psyllid also occurs in Canada (Alberta, British Columbia, and Saskatchewan) and in Mexico and Central America (Guatemala, Honduras, and Nicaragua) (Trumble 2009). In Washington and Oregon, severe colonization of the potato fields occurs in late June and early July (Crosslin et al. 2012). In Florida, the psyllid Bactericera cockerelli has been intercepted on poblano peppers from Mexico (Halbert and Dixon 2014)
Diagnosis
The most reliable method for the detection of the Ca. L. solanacearum is polymerase chain reaction (PCR) assay using species specific primers. However, the choice of the sample tissue used for the diagnosis is crucial for the successful detection of this bacterium using PCR. Ca. L. solanacearum is phloem-limited, so vascular tissue is the best sample source for successful PCR detection. Higher concentrations of the bacterium were observed in root, tuber, and stolon tissue than in the aerial tissue of potato plants (Li et al. 2009; Wen et al. 2009). In potato, yellowing or purpling leaves, the shoots of the infected symptomatic plants, and aerial tubers are most reliable (Australian Department of Agriculture 2011). For tomatoes, symptomatic shoot tissues, including yellow leaves, stems, leaf petioles, the peduncles attached to fruit, and the portion of affected fruit to which the peduncle is attached (Australian Department of Agriculture 2011), are the most reliable tissue source for diagnosis.
Management Strategies
Currently, all management strategies for the zebra chip and psyllid yellows diseases target the potato/tomato psyllid. Monitoring for the psyllid is essential for the effective management of the pest. Early-season detection and management of this insect is crucial to minimize psyllid reproduction and spread of the disease throughout fields.
Yellow sticky or water-pan traps can be used to monitor the presence and movement of potato psyllids in a given region (Al-Jabar and Cranshaw 2007). The use of neon-green traps is also reported to be highly efficient in monitoring the vectors.
Visual examination of the underside of the leaves may be required for sampling of the eggs and nymphs. The psyllid nymphs can be difficult to see on a potato plant, so planting a few sentinel bean or pepper plants, which have smooth leaves and are readily infested, can provide an easier way to detect the psyllids (Godfrey and Haviland 2008).
Treatment with imidacloprid in the potato field at the time of planting is an appropriate way to reduce the psyllid vectors (Godfrey and Haviland 2008; Butler, Walker, and Trumble 2012). Insecticides should be targeted to the specific growth stage of the vector. Depending on the severity of the infestation, additional treatments of the commonly used insectides for the potato psyllid may be recommended. These commonly used insecticides include acephate, metamidophos, buprofezin, abamectin, cypermethrin, lambda-cyhalothrin, esfenvalerate, spinosad, spirotetramat, imidacloprid, thiamethoxam, spiromesifen, dinotefuran, pyriproxyfen, and pymetrozine (Butler and Trumble 2012; Butler et al. 2011; Bynum, Henne, and Rush 2010; Godfrey and Haviland 2008; Zens et al. 2010).
Rotation of insecticides with different modes of action is recommended. In addition, good cultural practices, including removal of alternative hosts and solanaceous weeds and practices of on-farm biosecurity and hygiene, will help to prevent entry, establishment, and spread of the pests and diseases.
References
Abad, J. A., M. Bandla, R. D. French-Monar, L. W. Liefting, and G. R. G, Clover. 2009. "First Report of the Detection of 'Candidatus Liberibacter' Species in Zebra Chip Disease-infected Potato Plants in the United States." Plant Disease 93: 108.
Abdullah, N. M. M. 2008. "Life History of the Potato Psyllid Bactericera cockerelli (Homoptera: Psyllidae) in Controlled Environment Agriculture in Arizona." African Journal of Agricultural Research 3: 60–67.
Alfaro-Fernández, A., M. C. Cebrián, F. J. Villaescusa, A. Hermoso de Mendoza, J. C. Ferrándiz, S. Sanjuán, and M. I. Font. 2012a. First Report of 'Candidatus Liberibacter solanacearum' in Carrot in Mainland Spain. Plant Disease 96: 582.
Alfaro-Fernández, A., F. Siverio, M. C. Cebrián, F. J. Villaescusa, and M. I. Font. 2012b. "'Candidatus Liberibacter solanacearum' Associated with Bactericera trigonica-affected Carrots in the Canary Islands." Plant Disease 96: 581.
Al-Jabar, A. M., and W. S. Cranshaw. 2007. "Trapping Tomato Psyllid, Bactericera cockerelli (Sulc) (Hemiptera: Psyllidae), in Greenhouses." Southwestern Entomologist 32: 25–30.
Australian Department of Agriculture. 2011. "Diagnostic Protocol for the Identification and Detection of Candidatus Liberibacter solanacearum, the Causal Agent of Zebra Chip of Potatoes." Accessed June 07, 2015. http://plantbiosecuritydiagnostics.net.au/wordpress/wp-content/uploads/2014/09/NDP-18-Zebra-chip-Candidatus-Liberibacter-solanacearum-V1.1.pdf
Brown, J. K., M. Rehman, D. Rogan, R. R. Martin, and A. M. Idris. 2010. "First Report of "Candidatus Liberibacter psylaurous" (syn. "Ca. L. solanacearum") Associated with the 'Tomato Vein Greening' and 'Tomato Psyllid Yellows' Diseases in Commercial Greenhouse in Arizona." Plant Disease 94: 376.
Buchman, J. L., V. Sengoda, and J. E. Munyaneza. 2011. "Vector Transmission Efficiency of Liberibacter by Bactericera cockerelli (Hemiptera: Triozidae) in Zebra Chip Potato Disease: Effects of Psyllid Life Stage and Inoculation Access Period." Journal of Economic Entomology 104: 1486–1495.
Butler, C. D., and J. T. Trumble. 2012. "The Potato Psyllid, Bactericera cockerelli (Sulc) (Hemiptera: Triozidae): Life History, Relationship to Plant Diseases, and Management Strategies." Terrestrial Arthropod Reviews 5: 87–111.
Butler, C. D., F. R. Byrne, M. L. Keremane, R. F. Lee, and J. T. Trumble. 2011. "Effects of Insecticides on Behavior of Adult Bactericera cockerelli (Hemiptera: Triozidae) and Transmission of Candidatus Liberibacter psyllaurous." Journal of Economic Entomology 104: 586–594.
Butler, C. D., G. P. Walker, and J. T. Trumble. 2012. "Feeding Disruption of Potato Psyllid, Bactericera cokerelli, by Iimidacloprid as Measured by Electrical Penetration Graphs." Entomologia Experimentalis et Applicata 142: 247–257.
Bynum, E., D. C. Henne, and C. M. Rush. 2010. "Evaluation of Spray Applications for Potato Psyllid and ZC Management." In Proceedings of the 10 Annual Zebra Chip Reporting Session, edited by F. Workneh and C. M. Rush, 88–92.
Crosslin, J. M., and G. Bester. 2009. "First Report of Candidatus Liberibacter psyllaurous in Zebra Chip Symptomatic Potatoes from California." Plant Disease 93: 551.
Crosslin, J. M., and J. E. Munyaneza. 2009. "Evidence that the Zebra Chip Disease and the Putative Causal Agent can be Maintained in Potatoes by Grafting and in vitro." American Journal of Potato Research 86: 183–187.
Crosslin, J. M., P. B. Hamm, J. E. Eggers, S. I. Rondon, V. G. Sengoda, J. E. Munyaneza. 2012. "First Report of Zebra Chip Disease and "Candidatus Liberibacter solanacearum" on Potatoes in Oregon and Washington State." Plant Disease 96: 452.
Crosslin, J. M., N. Olsen, and P. Nolte. 2012. "First Report of Zebra Chip Disease and "Candidatus Liberibacter solanacearum" on Potatoes in Idaho." Plant Disease 96: 453.
Halbert, S. E., and J. E. Munyaneza. 2012. "Potato Psyllids and Associated Pathogens: a Diagnostic Aid." Florida State Collection of Arthropods, Gainesville, FL. Accessed June 07, 2015. http://www.fsca-dpi.org/Homoptera_Hemiptera/Potato_psyllids_and_associated_pathogens.pdf
Halbert, S., and W.N. Dixon. 2014. "Potato Psyllid (Bactericera cockerelli) (Hemiptera: Psyllidae) a Pest of Solanceae and Vector of Plant Pathogens Established in the Western USA." Pest Alert: Florida Department of Agriculture and Consumer Service, Department of Plant Industry. Accessed June 07, 2015. http://www.freshfromflorida.com/Divisions-Offices/Plant-Industry/Plant-Industry-Publications/Pest-Alerts/Potato-Psyllid-Bactericera-cockerelli-Hemiptera-Psyllidae.
Hansen, A. K., J. T. Trumble, R. Stouthamer, T. D. Paine. 2008. "A New Huanglongbing Species, 'Candidatus Liberibacter psyllaurous' Found to Infect Tomato and Potato, is Vectored by the Psyllid Bactericera cockerelli (Sulc)." Applied and Environmental Microbiology 74: 5862–5865.
Godfrey, L. D., and D. R. Haviland. 2008. Potato Psyllid. UC IPM Pest Management Guidelines: Potato. Accessed June 07, 2015. http://www.ipm.ucdavis.edu/PMG/r607300811.html#LINKS.
Liefting, L. W., Z. C. Perez-Egusquiza, G. R. G. Clover, and J. A. D. Anderson. 2008a. "A New 'Candidatus Liberibacter' Species in Solanum tuberosum in New Zealand." Plant Disease 92: 1474.
Liefting, L. W., L. I. Ward, J. B. Shiller, and G. R. G. Clover. 2008b. "A New 'Candidatus Liberibacter' Species in Solanum betaceum (Tamarillo) and Physalis peruviana (Cape Gooseberry) in New Zealand." Plant Disease 92: 1588.
Liefting, L. W., P. W. Sutherland, L. I. Ward, K. L. Paice, B. S. Weir, and G. R. G. Clover. 2009. "A New 'Candidatus Liberibacter' Species Associated with Diseases of Solanaceous crops." Plant Disease 93: 208–214.
Li, W., J. A. Abad, R. D. French-Monar, J. Rascoe, A. Wen, N. C. Gudmestad, G. A. Secor, I. M. Lee, Y. Duan, and L. Levy. 2009. "Multiplex Real-time PCR for Detection, Identification and Quantification of "Candidatus Liberibacter solanacearum" in Potato Plants with Zebra Chip." Journal of Microbiological Methods 78: 59–65.
McKenzie, C. L., and Shatters, R. G. 2009. "First Report of "Candidatus Liberibacter psyllaurous" Associated with Psyllid Yellows of Tomato in Colorado." Plant Disease 93: 1074.
Munyaneza, J. E., J. M. Crosslin, and J. E. Upton. 2007. "Association of Bactericera cockerelli (Homoptera: Psyllidae) with "Zebra Chip", a New Potato Disease in Southwestern United States and Mexico." Journal of Economic Entomology 100: 656–663.
Munyaneza, J. E., J. L. Buchman, and J. M. Crosslin. 2009. "Seasonal Occurrence and Abundance of the Potato Psyllid, Bactericera cockerelli, in South Central Washington." American Journal for Potato Research 86: 513–518.
Munyaneza, J. E., V. G. Sengoda, J. M. Crosslin, J. A. Garzon-Tiznado, and O. G. Cardenas-Valenzuela. 2009. "First Report of "Candidatus Liberibacter in Pepper Plants in Mexico." Plant Disease 93: 1076.
Munyaneza, J. E., T. W. Fisher, V. G. Sengoda, S. F. Garczynski, A. Nissinen, and L. Lemmetty. 2010. "First Report of "Candidatus Liberibacter solanacearum" in Psyllid-Affected Carrots in Europe." Plant Disease 94: 639.
Munyaneza, J. E. 2012. "Zebra Chip Disease of Potato: Biology, Epidemiology, and Management." American Journal of Potato Research. 89: 329–350.
Munyaneza, J. E., V. G. Sengoda, J. L. Buchman, and T. W. Fisher. 2012. "Effects of Temperature on 'Candidatus Liberibacter solanacearum' and Zebra Chip Potato Disease Symptom Development." Plant Disease 96: 18–23.
Rehman, M., J. Melgar, C. Rivera, N. Urbina, A. M. Idris, and J. K. Brown. 2010. "First Report of "Candidatus Liberibacter psyllaurous" or "Ca. Liberibacter solanacearum" Associated with Severe Foliar Chlorosis, Curling, and Necrosis and Tuber Discoloration of Potato Plants in Honduras." Plant Disease 94: 376.
Secor, G. A., V. Rivera-Varas, J. A. Abad, I. M. Lee, G. R. G. Clover, L. W. Liefting, X. Li, and S. H. De Boer. 2009. "Association of 'Candidatus Liberibacter solanacearum' with Zebra Chip Disease of Potato Established by Graft and Psyllid Transmission, Electron Microscopy, and PCR." Plant Disease 93: 574–583.
Trumble, J. 2009. "Potato Psyllid." Center for Invasive Species Research, University of California Riverside. Accessed June 07, 2015. http://cisr.ucr.edu/potato_psyllid.html
Wallis, R. L. 1955. "Ecological Studies on the Potato Psyllid as a Pest of Potatoes." USDA Technical Bulletin 1107.
Watson, C. 2009. "Industry Perspectives and Observations: Tamarillo Growers." Solanaceous Crops – Psyllids & Liberibacter Proceedings of the Workshop held 16 March 2009, Christchurch, New Zealand 7 World Potato Congress 50 –53.
Wen, A., I. Mallik, V. Y. Alvarado, J. S. Pasche, X. Wang, W. Li, L. Levy, H. Lin, H. B. Scholthof, T. E. Mirkov, C. R. Rush, N. C. Gudmestad. 2009. "Detection, Distribution, and Genetic Variability of 'Candidatus Liberibacter' Species Associated with Zebra Complex Disease of Potato in North America." Plant Disease 93: 1102–1115.
Yang, X. B., Y. M. Zhang, L. Hua, and T. X. Liu. 2010. "Life History and Life Tables of Bactericera cockerelli (Hemiptera: Psyllidae) on Potato Under Laboratory and Field Conditions in the Lower Rio Grande Valley of Texas." Journal of Economic Entomology 103: 1729–1734.
Zens, B., C. M. Rush, D. C. Henne, F. Workneh, E. Bynum, C. Nansen, and N. C. Gudmestad. 2010. "Efficacy of Seven Chemical Programs to Control Potato Psyllids in the Texas Panhandle." In Proceedings of the 10th Annual Zebra Chip reporting Session, edit by F. Workneh and C. M. Rush, 83–87.


