This publication is best viewed online in pdf format, https://edis.ifas.ufl.edu/pdffiles/AN/AN17100.pdf.
This section includes pages 11–36 of the complete handbook.
- Injection Site Management
- Best Management Practices - Injections
- Residue Avoidance
- Preventative Herd Health Plan
- Best Management Practices—Antibiotic Use
- Best Management Practices—Medicated Feeds
- Best Management Practices—Chemical Residues
- Best Management Practices—Feed Contaminants
- Best Management Practices—Ruminant By-products
- Best Management Practices—Beef Measles
- Best Management Practices—Feed Toxins
- Foreign Object Avoidance
- Best Management Practices—Birdshot/Buckshot
- Best Management Practices—Feed Records
- Best Management Practices—Broken Needles
All sections of the handbook are listed at https://edis.ifas.ufl.edu/collections/book_bsqa
Food Safety Control Points
These are called "food safety" control points because a legitimate food safety risk of sufficien tseverity exists to warrant control. Most cow-calf and stocker operations will only have a few management points that are truly food safety control points. The primary concerns associated with food safety are pathogens, residues (antibiotic/chemical) and foreign materials (buckshot/broken needles). It is imperative that food safety control points are identified so preventive and corrective measures can be put in place. Control points in cow-calf and stocker production that have potential consequences for the safety of beef are:
There may be other control points in a beef operation. It is important for you to develop your own production chart or list that includes all of the management practices you employ in your operation. That chart can then be used to identify your particular control points.
Managing food safety control points
Problems and concerns for food safety fall under three areas:
- Injection site management
- Residue avoidance
- Antibiotic residues
- Chemical residues
- Feed contamination and residues
3. Foreign object avoidance
Injection Site Management
Although most injection site management issues are concerned with quality control, improper administration of animal health products can lead to food safety risks. Injection site lesions and abscesses do not pose a health risk to consumers. However, consumers perceive they are health risks and, as the saying goes, their perception is our reality. So, quality defects caused by improper injection site management are a safety concern that should be addressed as a food safety control point. Problems like broken needles and violative residues can lead to adulteration of the carcass. These problems make the affected beef products unsafe for consumption. The following guidelines should be followed for both food safety control points aswell as quality control points (See more about quality control points on page 43).
Best Management Practices—Injection
- Follow instructions. Make sure you read the label and follow all labeling informationbefore administering any animal health product. Avoid intramuscular (IM) injections whenever other labeled routes of administration are available.
- Use proper restraint when administering injections to cattle. Improper restraint is the leading cause of broken needles and tissue damage.
- All IM injections, regardless of the animal's age, must be given in the NECK only–no exceptions! When administering subcutaneous (SQ) injections, use the "tenting" technique (See Figure 1), and when possible, place the injection in the neck. As a last resort, an acceptable SQ site would be behind the elbow in the elbow pocket.
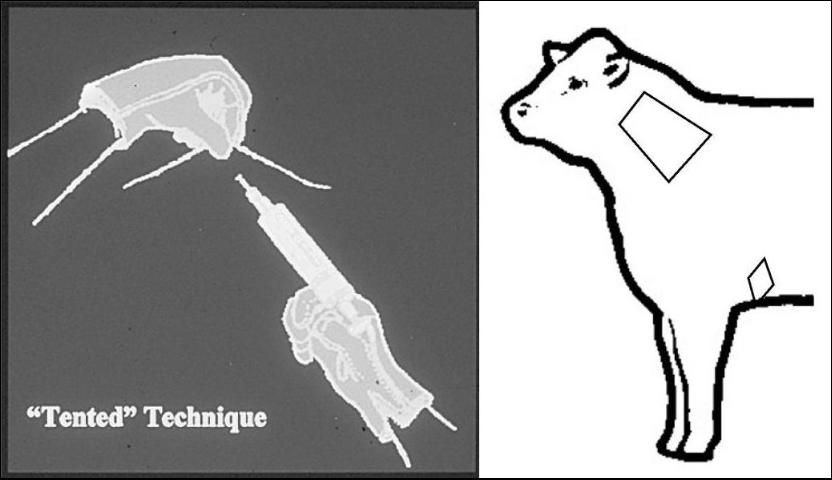
4. Use SQ, oral, intravenous (IV) or topical administration of antibiotics, vaccines and parasiticides when possible.
5. Never exceed 10cc in any injection site. For example, if 24cc is the recommended dose, use three 8cc injections instead of two 12cc injections.6.If possible, do not place more than one SQ injection on the same side of the neck to avoid interaction of products or severe tissue reaction.
6. If possible, do not place more than one SQ injection on the same side of the neck to avoid interaction of products or severe tissue reaction.
7. Properly space injections:
- 3 inches between injection sites on calves and yearlings.
- 4 inches between injection sites on cows and bulls.
8. Never mix products. Mixing products can cause unnecessary tissue damage, may reduce the effectiveness of the products administered or extend the withdrawal period before the animal can be sold and potentially go to a packing plant.
9. Use needles no larger than necessary. Proper needle size will vary depending on product viscosity, size of animal and route of administration (IM or SQ).
- 16–18 gauge ½- to 1-inch needles work well for SQ injections.
- 16–18 gauge 1- to 1 ½-inch needles work well for IM injections (See Table 1, Guidelines for Needle Selection).
10. Protect needles from contamination.
11. Change your needle when it becomes contaminated or damaged. Change needles frequently (10 to 12 head per needle) to ensure minimal tissue damage from burrs and minimize the risk of carrying contaminant into the injection site. Change needles on every animal if a blood-borne pathogen (ie. anaplasmosis) is known to exist in your herd. If a needle bends, stop immediately and replace it. Do not straighten it and use it again. Bent needles are much more likely to break off in the animal.
12. Injection sites should be free of soil and manure. Processing cattle in wet weather increases the chance of injection-site contamination.
13. Do not use chemical disinfectants to sterilize needles or syringes. To sterilize, boil syringe components and reusable needles in water for 20 minutes. Disinfectantscan cause severe tissue irritation and will reduce the efficacy of products like Modified Live Virus (MLV) vaccines. It is best not to disinfect the injection site as product contamination can occur, as well as increased tissue damage.
14. Develop a record-keeping system and processing map (See Record Keeping for BeefQuality Assurance, page 40) to document individual animals or entire groups ofanimals that have been treated. Also, include the route of administration used(IM or SQ), product used, product lot number and serial number (in the event you encounter an episode of product or treatment failure).


Residue Avoidance
Adulteration of beef products can occur with residues from animal health products, herbicides, pesticides and chemical contaminants of feed and water. Traces of some drugs and chemicals may be allowed in certain tissues. This is known as the tolerance level.
Tolerance levels are usually discussed in terms of one part of drug or chemical to one million or one billion parts of tissue. For some chemicals, no detectable amount is allowed (zero toler-ance). The Food and Drug Administration establishes tolerance levels for residues in food products.
Residues are monitored through sampling of meat products and suspect animals in beef processing facilities. Violations of the legal limits called violative levels can result in regulatory action, including fines, herd quarantine, and possibly criminal prosecution.
To date, violations have been minimal. But recent changes in inspection and monitoring may result in a higher incidence of residue detection.
Traces of some drugs and chemicals may be allowed in certain tissues. This is known as the tolereance level.
The Food and Drug Administration, the US Department of Agriculture and the Environmental Protection Agency approve and establish guidelines for the use of animal health products and agricultural chemical products used in pasture and range management, crop production, feed processing and storage.
During the approval process, withdrawal times are established for livestock treated with or exposed to regulated compounds and products. These times are explicitly defined on the labels for the products. The first step in avoiding residues is to read and follow label directions for all products used in beef and other agricultural production. In addition to animal health products and pasture and range pesticides, contamination or residues may result from accidental or negligent exposure to feed, water, or soil that has been contaminated with heavy metals, petrochemicals, PCBs, PCPs, insecticides, fungicides, herbicides, mycotoxins, or other hazardous materials.
Careful management and oversight is necessary to prevent exposure to these compounds.
Residue Monitoring in Non-Fed Cattle (Cull Cows/Bulls)
Residues in fresh meat and poultry are monitored by the Food Safety Inspection Service through the National Residue Program (NRP). The NRP helps prevent the entry of animals containing violative residues of pesticides, drugs or potentially hazardous chemicals into the food chain through monitoring and enforcement.
Random samples are tested for monitorin gthe national residue incidence. Specific samples are collected for enforcement based on clinical signs and previous herd history.
Traditionally, animals were selected for testing based on pre-harvest evaluation only (down, disabled, recent surgery). Effective Aug. 9, 1999, inspectors were instructed to check for residues after harvest in animals with any of the following 12 conditions:
- Downers
- Suspects
- Mastitis
- Pneumonia
- Body-cavity lining inflammation
- Heart sac lining inflammation
- Skin inflammation
- Twisted stomach disease
- Septicemia (blood poisoning)
- Pyemia (blood poisoning)
- Injection sites
- Uterine infection
Violations of the legal limits calledviolative levels can result in regulatoryaction, including fines, herd quarantine, and possibly criminal prosecution.
In cull cows and bulls, residues are monitored and can be traced back to the owner through back tags that are applied at the auction market or packing plant. In 1997 (the most current year reported), the majority of violative residues for antibiotics and sulfonamides occurred in tissue samples from dairy cows. But, beef cows were also a significant source.
Based on results from the 1999 Market Cow and Bull Beef Quality Audit, each carcass tested for residues costs nearly $45. That averages out to almost one dollar ($0.92) for every non-fed animal marketed that year.
The impact may seem small on a per cow basis, but nationally there areapproximately 6 million cull cows/bulls harvested every year. As well, the cost of inspecting for residues in the end product goes against the principlesof HACCP and TQM, which stress prevention rather than inspection.
These problems can and must be solved at the producer level, and progress in reducing residues will only be accomplished if producers pay strict attention to guidelines for proper use of animal health products and other potential contaminants.
Avoiding Antibiotic Residues
Overall, the beef industry is doing an excellent job of controlling violative drug residues by placing emphasis on the identification and handling of individually treated cattle. This includes identifying each animal treated, accurately recordingt he treatment, date and treatment dose, and following prescribed withdrawal times.
It is important that beef producers establish a working relationship with a licensed veterinarian. Find and use a veterinarian who is willing to be involved with your Beef Quality Assurance program. Be cautious about cattle treatment advice from anyone who is not highly acquainted with your operation and the proper use of animal health products.
Based on results from the 1999 Market Cowand Bull Beef Quality Audit, each carcass tested for residues costs nearly $45. That averages out to almost one dollar ($0.92) for every non-fed animal marketed that year.
Treatment Protocol Book
Ask your veterinarian to help you develop a "treatment protocol book" specific to your operation. While this concept may be more familiar to feedyards and larger stocker operations, it's also avaluable management practice for cow-calf producers. In simple terms, it involves writing down a plan for what treatment(s) to use when cattle get sick for various reasons.
You'll also need to write down your plan for follow up and/or alternative treatments if the initialtreatment doesn't produce the desired result. A treatment protocol book should be reviewed regularly and updated as often as appropriate (as newi nformation or products become available, if your planned treatments aren't working, etc.).
Keep the treatment protocol book on file in your home or office and at your working facilities. As you update it, all previous versions should be kept on file for a year or more, so that you can refer back to treatments that have worked in previous situations. Whenever you update your book, it must have the veterinarian's signature and date recorded when the book was reviewed. Listing products that have a withdrawal time in your treatment protocol book is a Best Management Practice (BMP).
Overall, the beef industry is doing an excellent job of controlling violative drug residues by placing emphasis on the identification and handling of individually treated cattle.
Preventative Herd Health Plan
The most effective way to reduce the potential for antibiotic residues is to control the need to use them. Every effort should be made to prevent disease and infection in the cattle herd. To accomplish this a herd health plan needs to be developed for each individual ranch operation. One herd health plan will not fit all operations across the state.
Preventative herd health plans will consist of herd management and immunization recommendations. Work with your veterinarian to develop an annual herd health program. Included in this plan should be:
- Targeted pathogens.
- Recommended vaccines.
- Recommended feed additives (if any).
- Appropriate time frame to protect (vaccinate) against targeted pathogens.
- Additional management considerations to aid in the prevention or spread oftargeted pathogens.
- Development of management and treatment protocols in the event of failed prevention efforts. This will require an outline of treatment protocols by yourveterinarian.
Management and treatment considerations will need to be discussed and developed on and for each operation. You and your veterinarian will need to develop the treatment protocols so that you are both comfortable with the recommendations. The preventative herd health plan, treatment protocols and veterinary drug orders need to be developed together to complete your herd health program.
Any medication that requires a use other than as directed on the label must have revised administration procedures included in the treatment protocol book.Your veterinarian should supply a revised label including the veterinarian's name, address, phone number, revised directions for use, name of drug and withdrawal time.
Drugs licensed and distributed for preventing and treating of diseases and parasites, increasing weight gains, synchronizing estrus and other uses have specific label instructions. These include the period of time that must pass after the last dose is given until harvest of the animal.
This period of time is known as the withdrawal period and is usually stated in hours or days. The withdrawal period stated on the label allows time for elimination of the drug from the animal, or reduction of drug residues to below tolerance levels before slaughter.
Extra-label drug use is using a drug at a dose, by a route, for a condition or indication, or in a species not on the label. Extra label use is not legal for performance enhancement.T o use a drug "extra-label" requires a veterinary prescription. If no prescription is obtained them "extra-label"drug use is referred to as "off-label" and is illegal!!
Extra-label drug use requires a revised withdrawal time established by the prescribing veterinarian. This results in extended withdrawal periods in treated animals in order to reduce the leve lof residues below violative levels. Withdrawal times may also be extended for animals that have been severely impaired by disease, malnutrition or age.
Avoiding tissue residue of antibiotics is simple to manage. Observe and follow label directions and ensure that cattle are not marketed until the appro-priate withdrawal time has elapsed. On the next page are basic management practices necessary to assure that no violative antibiotic residues will be present in carcass tissues.
Withdrawal period: the period of time hat must pass after the last dose is given until harvest of the animal. The withdrawal period stated on the label allows time for elimination of the drug from the animal, or reduction of drug residues to below tolerance levels before harvest.
Best Management Practices—Antibiotic Use
- Strictly follow all recommendations and guidelines from your veterinarian for selection of products.
- Follow label directions for use of the product. Use the product at recommended dosage for required time period. Treatment regimens must comply with label directions unless otherwise prescribed by a veteri-narian. If drugs are to be used in an extra-label manner, that must be done under the prescription or direct supervision of a licensed veterinarian. (The requirements for a valid veterinarian-client-patient rela-tionship (VCPR) are covered in the Appendix, page 101.) All cattle treated in an extra-label manner must comply with prescribed withdrawal times, which have been set by your veterinarian under the guidelines of a valid VCPR. The Florida Beef Quality Producer program does not support/recommend extra-label drug use (ELDU) for injectable aminoglycosides (such as neomycin, gentamicin or kanamycin) because of the potentialviolative residues related to extremely long withdrawal times. Some studies have shown withdrawal times on these types of products could be as long as 18 months.
- Accurately calculate dose requirements based on the animal's weight and the specific health problem being treated. Providing the same drug simultaneously by injection, feed, or water may result in over dosing and, thereby, create a residue problem.
- When administering injectable products, follow the Best Management Practices—Injections outlined on page 13.
- Never administer more than 10cc per injection site. Exceeding this amount will increase tissue damage, alter withdrawal time and may require testing before cattle are marketed for consumption.
- Do not mix products prior to administration. This practice of using "bloody mary" mixes is compounding use and will result in undetermined withdrawal periods.
- All animals treated for problems unique to the individual animal should be recorded by the animal's ID, treatment date, disease indication, drug, and dose administered, product serial/lot number, approximate weight of animal, route and location of administration, and the earliest date the animal would clear the prescribed or labeled withdrawal period. (See page 39 and 40 for sample treatment records). You can record treatments either by individually identifying each animal in your herd and/or individually identifying each animal when or if they are treated. The ID number should be unique to that animal and tie it to the group from which it came.
- A special note for producers who do not individually identify animals.Identifying each animal individually is not required to participate in this program. Cattle can be identified by group. However, if treated cattle are not individually identified, then the entire group must be managed together until the appropriate withdrawal times have elapsed for every animal in the group. The withdrawal time applies to the entire group of animals. For example, let's say several calves develop scours and numerous calves are treated within a 10-day period. The entire group of calves would receive a withdrawal date based on the last date of administration of the product (to any individual animal) with the longest withdrawal period.The complete history of product use should be available for transfer when the group of cattle is sold or moved to the next production unit within your operation. Otherwise, your buyer (or the foreman of your other unit) will not be aware of when those calves can safely enter the marketing chain. For example, when a stocker operator culls his non-performing steers any time during the course of a grazing period, those animals could potentially be sent to a packer. If the stocker operator is unaware that the prior owner treated the animal with an antibiotic whose withdrawal time has not expired, he might have unknowingly contributed to a violative residue problem.
- All animals treated as part of a group will be identified by group or lot with treatment information recorded. Records should include the animal lot or group identification, processing/treatment date, product serial/lot number,product and dose administered, route and location of administration and withdrawal information. Recording animals under this system assumes that every animal in the lot or group received the treatment. (See forms forrecording group treatment history on page 40).
You DON'T know where they are going.
Any animal marketed from a cow-calf or stocker operation could potentially go into a meat product. You may sell an animal with no intent of it going to slaughter; however, the person you sold the animal to could resell it within a matter of days to someone else who sends it to slaughter. This applies to cows, bulls, calves and yearlings. That's why it is so important to observe withdrawal times whenever you sell cattle.
10. All cattle marketed from the ranch can potentially go directly to slaughter. Therefore, records for any cattle to be marketed should be checked by ranch personnel to ensure that treated animals will meet or exceed label withdrawal times for all products administered. A release slip should be signed and dated by the person who checks records prior to shipping cattle from the operation. The examination should include processing records, feeding records, treatment records and all otherrecords that may apply.
11. Extended withdrawal times should be expected for emaciated or severely debilitated animals. All cattle sold that are not typical of the herd (medicated cull cows and realizer cattle) may be subject to verification of drug withdrawal. (Realizers are animals with a health problem that get culled because they never recover.) Should there be any question about withdrawal period, the veterinarian will evaluate the treatment history against information provided by the Food Animal Residue Avoidance Databank (FARAD), and the animal may have to pass aresidue screening test, such as the Live Animal Swab Test (LAST), which tests for antibiotic residues. Residue screening will be performed by qualified personnel under the supervision of a veterinarian. The results will determine whether the animals can be released for shipment, but cannot be used to shorten the labeled withdrawal time. Attempting to salvage sick animals by treatment and prompt slaughter requires an accurate diagnosis and careful selection of drugs.
12. Make sure that all employees are aware of the proper use and administration of antibiotics and withdrawal times, and they have the ability to check appropriate withdrawal restrictions before moving cattle to market. For example, you can provide your employees with charts or software to help them track withdrawal dates.
Feed Additives and Medications
The term "medicated feed" includes all medicated feed products intended to be a substantial source of nutrients in the diet of an animal. The term includes products commonly referred to as supplements (medicated mineral fed to prevent anaplasmosis), concentrates (grain mixture that contains med-ication), premix feeds (concentrated medications mixed with additional roughage or concentrates), and base mixes, and is not limited to complete feeds (preconditioning chow for used at receiving/weaning).
For more detail on FDA regulations concerning feed additives and medicated feeds, see Appendix, page 103. In addition to the considerations listed above, the following recommendations relate specifically to the use of medicated feeds.
Best Management Practices—Medicated Feeds
- Only FDA-approved medicated feed additives can be used in rations.
- Feed only at recommended rates. Exercise caution when calculating rates for medicated feeds.
- All medicated feed additives will be used in accordance with the FDA-approved label. Extra-label use of feed additives is strictly prohibited by federal law. No one has the authority to adjust the dose as labeled, including veterinarians. All directions for the use of a medicated feed additive will be on the label attached to the bag or will be supplied with a bulk order. Water medications are not considered feed medications; therefore, they can be used under the extra-label drug use guidelines provided by the Center for Veterinary Medicine.
- Ensure that all additives are withdrawn at the proper time to avoid violative residues.
- For operations formulating and mixing rations on site, such as stocker operators, medicated feed additives will be used in accordance with the FDA Good Manufacturing Practices (GMPs). These include a formula record of all medicated feed rations produced and production records of all batches of feed produced that contain medicated feed additives. Feed production records must include additive used, date run, ration name, or number, the name of the person adding the additive or responsible for mixing the feed and amount produced. Use separate mixers for mixing medicated feeds and non-medicated feeds, or clean mixers between batches of each.
- Pre-mixed or formulated supplements typically used by many smalle rbeef operations and most cow-calf operations do not require FDA registration of any type. Larger beef operations that use certain highly concentrated medications may be required to register with the FDA via a FDA-1900 permit.
- Identify medicated-feed treated individuals or groups as described in the antibiotic use section, page 24.
Avoiding Chemical Rsidues
Pesticide or herbicide residue is not a major problem in the beef cattle industry, but it should be monitored in two main areas, products applied to he land and products applied directly to the animal. A third area of concern would be accidental or negligent exposure to feed, water, soil or other materials contaminated with hazardous materials. To avoid potential risk of residues, the following guidelines are recommendated.

Best Management Practices—Chemical/Pesticide Residues
- Use only agricultural chemicals approved for application to land grazed by livestock or on land where feedstuffs/hay are removed for animal consump\tion at a later time.
- Follow label directions and observe grazing restrictions on pastures, range-land and crops treated with pesticides.
- Prevent accidental exposure to agricultural chemicals by proper storage and disposal of containers. Do not use the same sprayer to apply agricultural chemicals to pasture or rangeland that you use to apply livestock pesticides directly to cattle.
- Only use products approved for control of internal and external parasites of cattle. Caution should be exercised when using petrochemicals, such as motor oil or diesel, in backrubbers or other self-treatment devices for control of external parasites. These compounds are routinely screened at harvest and overexposure can result in violative residues.
- Apply topical, oral and/or injectable livestock pesticides at label dose rate. Overdosing constitutes extra-label usage with unknown withdrawal times. Individual animal weights can help determine appropriate calculation of doses.
- Document usage and observe all appropriate withdrawal times before marketing cattle. Remember that residue problems occur more frequently with cullcows/bulls and realizer cattle than for healthy calves or yearlings.
- Prevent consumption of hazardous chemicals and heavy metals by proper storage and disposal of paint, batteries, chemical containers, used petrochemical products and other materials, and make sure cattle don't have access to petrochemical production sites.
- Prevent contamination of feedstuffs by chemical compounds through proper storage of chemicals and proper treatment of stored feed products with insecti-cides and fungicides. These should not be stored in the same location as approved animal-use products.
- Record dates of application, areas, animals and/or feedstuffs treated, products used, product serial and lot numbers, appropriate withdrawal periods, etc. Producers may request a letter of guarantee from the feed supplier that the feed is below violative levels for residues and mycotoxins. More pesticide information can be found at Florida Department of Agriculture and Consumer Services Bureau of Pesticides and the following websites:
dep.state.fl.us/mainpage/sitemap.htm
Feed Contamination
The potential for adulteration of beef from contaminated feed is greater than most producers realize. However, contamination is not common at the ranch level. Accidental contamination is much more common than any other type of problem. EPA pesticide product registration and licensed pesticide applicator requirements provide significant protection from pesticide residues in the US feed grain supply. In addition, costs associated with pesticides do not encourage over-application.
To make sure you do not buy a residue problem along with a load of manufactured feed, grain, by-products or crop residues, deal with a reputable feed commodity supplier. In addition, you may wish to ask suppliers about their use of grain protectants during storage and their monitoring procedures.
Fluid Leakage and Other Potential Contamination
The leakage of transmission and transformer fluid poses a potential problem in residue avoidance. Both types of fluid contain polychlorinated hydrocarbons (PCBs), which can leave a violative residue in cattle. While the occurrence of PCB residue from this source is small, the possibility still exists.
Another potential problem is transmission/hydraulic or radiator fluid that leaks from farm equipment and contaminates the feed. Lead and other heavy metals may be picked up through spills and leaks; batteries, paint and other materials may inadvertently contaminate feed or be picked up elsewhere by cattle.
Products used for bird and rodent control are another potential problem. While no residues have been reported from these products, they are toxic substances. While the chance of these products entering the feed source is small, care needs to be taken. Adhering to the guidelines on the next page can reduce the risk of residues from contaminated feed.
Best Management Practices—Feed Contaminants
- Maintain a quality control program for incoming feed ingredients in anattempt to eliminate contamination resulting from molds, mycotoxins, chemicals, and other contaminants.
- Store feed in a manner that prevents development of molds and mycotoxins and exposure to chemicals and other potential contaminants.
- Prior to usage, submit any feed ingredient suspected of contamination for analysis by a qualified laboratory.
- To avoid accidental livestock exposure, treat all chemicals as potential hazards. Never store chemical products where leakage or breakage can contaminate feed products or where cattle can directly consume a contaminant. For example, don't store batteries, fuel containers or paint in the same location as feedstuffs.
- Regularly check all feed-handling equipment for fluid leaks.
- Spills should be cleaned up to prevent potential contaminants from causing residues or death in cattle.
- If a feed-related poisoning is suspected, it is critical for the producer or veterinarian to contact a diagnostic laboratory for assistance in confirming the suspicion.
- If purchasing fats and oils, monitor for potential contamination. Letters of guarantee from companies supplying these materials may be requested that state these materials have been tested.
Ruminant By-Products
As of 1998, federal regulations prohibit the feeding of certain mammalian protein sources. The regulations primarily impact the feeding of meat meal and bone meal derived from ruminants. Tallow, blood by-products, gelatin, and milk products are excluded by the regulation and are acceptable for use in ration formulations. (More information on ruminant-derived by-products and their use is available in the Appendix, page 107.)
Best Management Practice—Ruminant By-products
- Do not use ruminant-derived protein sources in manufacturing ruminant feeds.
- Do not use other feed stuffs that may contain ruminate-derived proteins(i.e. poultry feed).
Beef Measles
Occasionally, feeders are notified by packers that some of their cattle have "measles." Cysticerosis, or "beef measles," refers to the immature larvae stage of the human tapeworm found in the form of cysts in the muscles of cattle. It results from cattle consuming feedstuffs contaminated with tapeworm segments or eggs, or cattle coming in contact with water or ground that has been contaminated by infected humans. If a worker defecates in a feedbunk, the feed can be contaminated with tapeworm segments or eggs. However, the tapeworm segments or eggs can also be shed from an infected human into the environment without defecating.
USDA regulations prohibit carcasses from being approved for human consumption if even one dead larva is found, unless the carcassis first subjected to refrigeration or heat sufficient to destroy all larvae. If cysts are distributed through out the carcass, it is condemned. Generally, only about 5 percent of carcasses with measles are totally condemned. The offal is always condemned. And because they have to freeze the carcasses and condemn the offal, packers significantly discount carcasses affected by measles. If laboratory reports indicate live cysts or more than 10 percent of the cattle in a feedyard are infested, USDA veterinarians investigate the premise of origin. Previous investigations have indicated that the majority of cattle with measles were infected prior to entering the feedyard.
Best Management Practices—Beef Measles
- Avoid high-risk feed sources.
- Observe septic leach fields and fix any broken pipes.
- Do not spread raw sewage onto pasture.
- Educate workers about the importance of personal hygiene near feedstuffs or feedbunks, water tanks or even pens where cattle could come in contact withtapeworm segments or eggs spread by infected humans.
Potential Microbial Contamination
As the beef industry strives to produce a safe and wholesome product, many areas of quality assurance take on new importance. Contamination of beef with various organisms of importance in human health is an increasingly grave concern. Recognized pathogens, such as E. coli 0157H7, Listeria spp. (all species) Salmonell aspp.and Campylobacter, may enter the beef supply in a number of ways.
While we do not have adequate methods today to eliminate microbial contamination in cattle production, attention to basic sanitation practices and proper animal health techniques can decrease the chance of microbial contamination.
Potential Feed Toxins
Mycotoxins are naturally occurring chemicals produced by fungi. Mycotoxins can be found in grains and forages, and if present in sufficient concentrations, can cause reduced feed consumption, poor production and adverse health effects that may result in residues in meat and milk products. Environmental conditions that are conducive to the growth of fungi and the production of mycotoxins are quite variable. Mycotoxins can be produced in feed-stuffs prior to harvesting or during storage and primarily occur in grain, peanuts and cotton by-products. Stress during critical stages of crop development often leads to aflatoxin development.
Best Management Practices—Feed Toxins
- Store feedstuffs in a manner to prevent mold formation and avoid feeding moldy feed.
- Maintain a quality control program for incoming feed ingredients in an attempt to eliminate contamination. It is important to keep in mind that mycotoxins can be present in feeds without visible mold growth; conversely, visibly moldy feed may not always contain detectable mycotoxins.
Foreign Object Avoidance (Adulteration)
There are two major types of foreign objects to be concerned with:
- buckshot or birdshot; and
- broken needles. On rare occasion, rifle bullet fragments and arrow tips have also been found in carcasses.
Birdshot/Buckshot
Lead birdshot/buckshot cannot be detected by metal detection devices used in packing and processing facilities. Furthermore, lead is considered an adulterant by the Food and Drug Administration. If the shot is detected on the slaughter floor, the entire carcass is routinely condemned.
If shot is detected during ground beef production, the entire lot of ground beef must be condemned. In large slaughter and processing plants, this can be several thousand pounds in one batch! In the1994 audit of non-fed beef cattle (cullcows and bulls), one processor commented that his company lost a total of 100,000 pounds in a six-week period due to the presence of lead shot.
In 1999, the Market Cow and Bull Beef Quality Audit revealed more than 10,000 head of non-fed slaughter cattle were condemned due to the presence of lead shot. In fact, the presence of buckshot/birdshot ranked third on the list of packer concerns; only bruises and antibiotic residues ranked higher.
Cattle producers tend to point their fingers at hunters. However, there are producers that sometimes use shotguns to gather unruly cattle. Regardless of who is at fault, this defect should be prevented with education about the consequences. Other means of animal control and capture must be used. To ensure that foreign objects are not found in carcasses, adhere to the following guidelines on the next page.

Best Management Practices—Birdshot/Buckshot
- Never use a shotgun to gather cattle. Develop alternative methods to control and capture animals. If an unruly animal cannot be trapped or gathered by some other means when it reaches the end of its productive life, the animalshould be euthanatized on the premise and disposed of properly (in otherwords, when you would normally cull the animal).
- Work with hunters to prevent shooting cattle with any weapon. Educate hunters to the potential safety concerns associated with adulterated carcasses. Remove cattle from hunting areas when possible to avoid accidental shootings.
Broken Needles
You and your veterinarian must determine how animals will be handled if a needle breaks off in the muscle when giving an injection. A broken needle is an emergency and time will be of the essence. Broken needles migrate in tissue, and if not handled immediately, the needle fragment will be impossible to find, requiring the animal to eventually be destroyed rather than sold at market.
Under no circumstances can animals carrying broken needles be sold or sent to a packer. Purchasing high-quality needles, changing and discarding damaged needles and providing proper restraint are all preventive measures.
Best Management Practices—Broken needles
- Restrain animals properly and adhere to injection site management as outlined on page 13.
- Do not straighten and use bent needles again. Replace immediately.
- Develop a standard operating procedure for dealing with needles broken off in cattle.
- If the needle remains in the animal, mark the location wherethe needle was inserted.
- If a broken needle cannot be removed at the ranch, contact a veterinarian immediately to have the needle surgically removed.
- If a broken needle cannot be extracted from the tissue, record the animal's ID to ensure that it is never sold or leaves the ranch. At the end of its productive life, the animal should be euthanatized and disposed of properly.