What are pentastomes?
The pentastomes are a group of worm-like parasites that infect many different species of fish. Infections have been found in several families of fish including the Cichlidae (tilapia), Cyprinidae (danios), Cyprinodontidae (flagfish), and Poecilidae (mosquitofish, swordtails, mollies, platies).
Although pentastomes are small, they can be seen without the use of a microscope. However, a microscope will help to differentiate them from other similar parasites. Pentastomes in fish resemble very small white, segmented grubs or worms (Figure 1). However, they are more closely related to fish lice such as Argulus than to true worms. They are believed to be close relatives of members of the group of organisms known as Crustacea, which include crabs, lobsters and shrimp, as well as the parasitic fish louse. Adult pentastomes, which are found in reptiles, look different from the larval or nymph stage, found in fish. They have been described as resembling "prehistoric caterpillars".
Typical pentastomid lesions seen on fish grossly resemble infections by digenean trematodes or flukes (see UF/IFAS Fact Sheet FA-28, Monogenean Trematodes). Digenean trematodes and pentastomids have different life cycles and different control methods.Therefore, proper identification is important. Light microscopy is a useful tool for distinguishing between the two.
Pentastome Life Cycle
Pentastomes have a complex, indirect life cycle (Figure 2). Infections cannot be transmitted directly from fish to fish. Adults of the pentastome species that infect fish are found in reptiles, which are considered to be their final hosts. Typically, adult stages of the parasite are found in the respiratory systems of reptiles, usually within the lungs and/or trachea (windpipe).
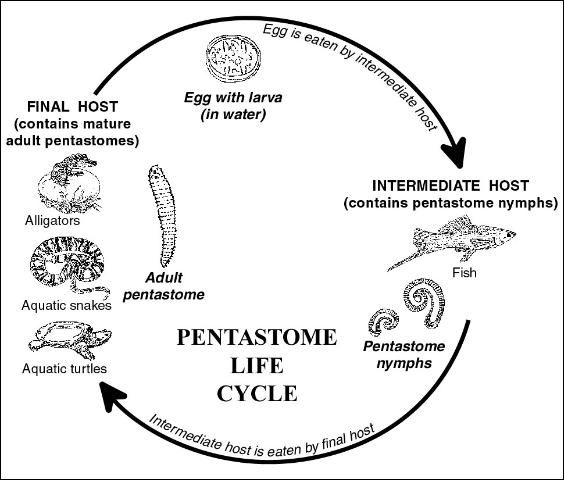
Within the lungs of their reptile hosts, the adult pentastomes deposit eggs containing larvae having four leg-like appendages. After deposition, these eggs are coughed up, swallowed, and then passed through the reptile's gastrointestinal tract where they are deposited into the water through the feces. The eggs develop into the infective stage and, after being eaten by the appropriate fish host, the larvae hatch out, develop, and undergo several molts. The juvenile life stage located in the fish more closely resembles a worm and is known as a "nymph". After infected fish are eaten by the proper final reptile host, the parasite perforates the intestinal wall, migrating through the body to the lungs. When the pentastome matures, the life cycle is completed.
A number of common reptiles are known reservoirs of pentastomes. Florida soft-shell turtles (Trionyx spp.) have been found harboring adult stages of one group of pentastomes in the genus Alofia. Alligators harbor adult pentastomes of the species Sebekia mississippiensis. Other species of pentastomes have been found in snapping turtles and aquatic snakes including the cottonmouth, red-bellied water snake, diamondback water snake, and the common water snake.
Disease in Fish Caused by Pentastomes
The appearance of the fish, as well as the amount of tissue damage done by the parasite, will depend on the fish species infected, whether or not the fish has been infected before, and the species and number of pentastomes present. In one published report (Boyce et. al. 1987), little damage was seen in infected mosquitofish, whereas the same species of parasite caused significant damage and inflammation in infected swordtails. Necropsy of the swordtails revealed several clinical findings: prominent swellings of the skin, tracks where parasites had migrated through muscle, and encapsulated juvenile (nymph) stages present in the body cavity as well as under connective tissue of many organs.
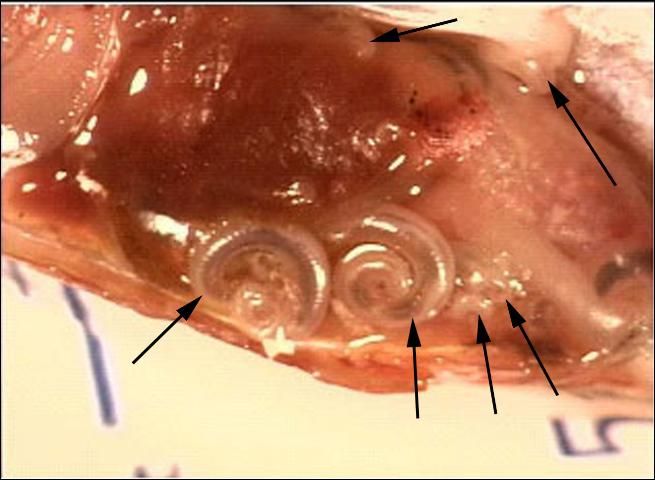
In some infected fish examined at the UF/IFAS Tropical Aquaculture Laboratory, there were obvious swellings in the skin and muscle from which nymph stages were extracted. Conversely, other fish had relatively few external signs of the parasite, however, in some of these fish, significant numbers of parasites were seen internally during necropsy (Figure 3). One swordtail had over 200 pentastomes of various sizes located throughout its body.
Diagnosis and Treatment
Affected fish may have grub-like lesions within the skin and muscle, however, infections can occur within internal organs of fish with no external signs present.
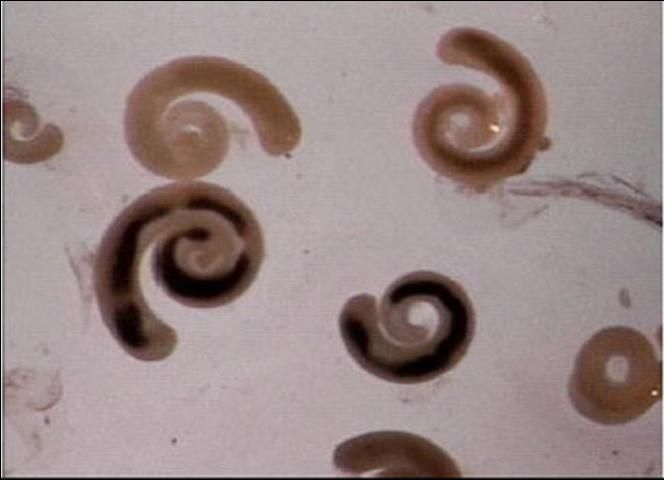
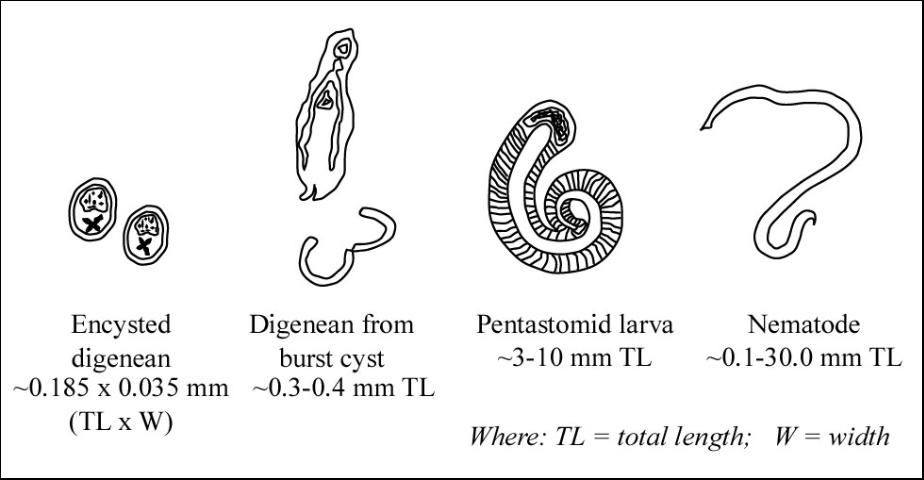
Common characteristics of pentastomes (Figure 4) include: a short body length relative to body diameter (compared with nematodes); obvious segmentation (annuli) of the body (nematodes are not segmented); small hooks on the anterior (head) end; and a coiled nymph stage. In contrast, digenean trematodes in fish are usually found as immature metacercariae (a larval stage in an oval, encysted form). If these cysts are broken, the digenean trematodes that emerge appear flatter and have circular oral and ventral suckers.
Because pentastomes can be found encapsulated in many different locations within fish, there are no treatments other than prevention. Chemical treatments to kill stages in the water have not been evaluated. Affected fish typically must be culled. Depending upon the severity of infection within a population, anywhere from a few percent to as high as 30 percent of the fish in a pond may be affected. Consequently, it is important that infections are recognized early so that preventative measures can be instituted as soon as possible.
Prevention
Primary prevention of pentastomid infections in fish requires control of the final hosts: aquatic turtles, water snakes, and alligators. Fish in ponds with any of these predators may be susceptible to pentastomid infections. Predator control should be standard practice in an aquaculture facility. Prolonged exposure to infected reptiles will increase nymph loads in susceptible fish by increasing the number of infective larvae in the ponds. This number will increase with time. Consequently, farms that have aquatic reptiles and do not regularly clean ( muck/pump down, rinse, and lime) and restock their ponds will be at greater risk of infection.
Discussion
Fish with whitish bumps or raised areas that contain parasites that resemble grubs should be examined by a fish health specialist for proper identification of the parasite. Some species of fish have significant disease from pentastomid infections that can contribute to morbidity or mortality. Other species of fish suffer no ill effects and may show no grossly visible signs of disease at all. Several different parasites or other diseases may cause a similar appearance on the fish, so an important first step is positive identification of the problem.
If pentastomes are determined as the cause, turtle, alligator, and water snake populations within the affected ponds or farm should be controlled and ponds cleaned and restocked. There are no known methods for chemical removal of pentastome parasites from infected fish, which consequently must be culled.
Reference
Boyce, W.M., Kazacos, E.A., Kazacos, K.R. and Engelhardt, J.A. 1987. Pathology of Pentastomid Infections (Sebekia mississippiensis) in Fish. Journal of Wildlife Diseases, 23(4): 689-692.