Molluscan Shellfish Aquaculture Worldwide
In the past five decades, global fisheries and aquaculture have grown steadily, and seafood consumption per capita has increased from an average of 9.9 kg in the 1960s to 19.2 kg in 2012 (FAO 2012). This development is impressive, and probably driven by multiple factors, such as population growth, rising income, and urbanization (Delgado et al. 2003; Diana 2009). Molluscan shellfish has traditionally been a major component of world aquaculture. For example, the molluscan shellfish production in 2012 (15.2 million metric tonnes; 1 metric ton equals to 1000 kilograms or 2204 pounds, hereinafter the same below) accounted for about 22.8% of the total (inland and marine) aquaculture production and 60.3% of the world marine aquaculture production (FAO 2014).
Worldwide, the major aquaculture molluscan species groups include clams (together with cockles and arkshells), oysters, mussels, scallops, abalones, and other miscellaneous molluscs. Based on their production in 2012, clams, oysters, mussels, and scallops were the four major groups and accounted for 87% of the total molluscan aquaculture production (Figure 1).
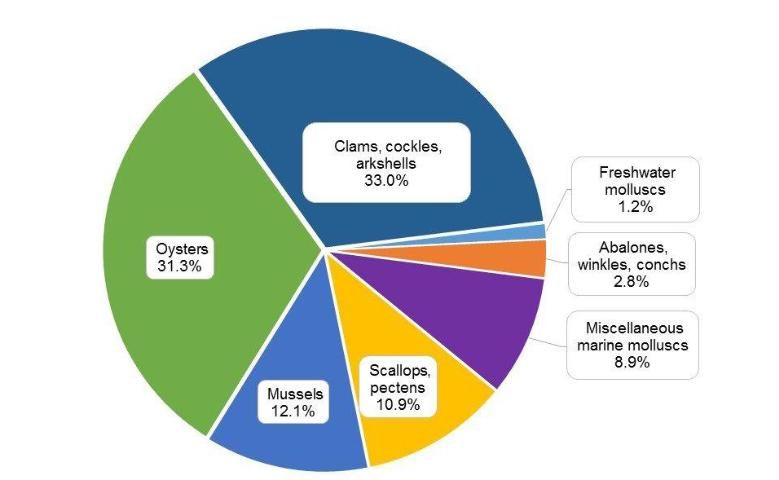
Credit: FAO 2014
The annual production (total weight) and sales values of the four primary molluscan aquaculture groups varied from year to year in the past decade (Figure 2 upper). Oysters led annual production between 2003 and 2007 but have been replaced by clams since 2008 (Figure 2 upper). Clams represent the leading product in sales value, at about 5.0 billion USD in 2012, followed by oysters, scallops, and mussels (Figure 2 lower).
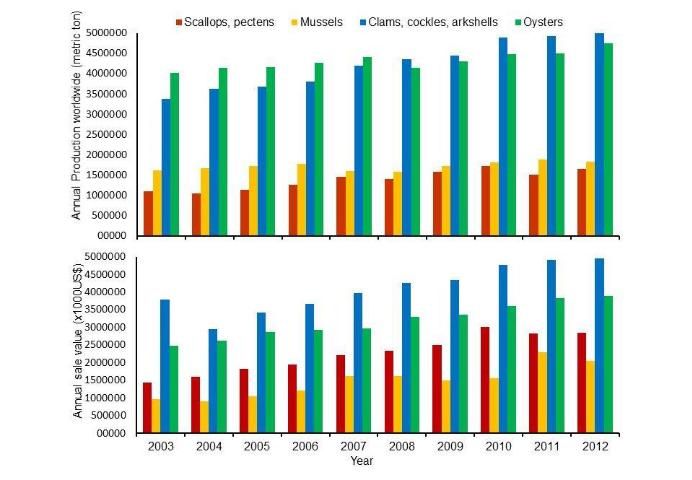
Credit: FAO 2012
Molluscs are cultured in 76 countries worldwide (FAO 2014). The top ten countries for aquaculture production are China (81.4%), followed by Vietnam (2.6%), Korea Republic (2.5%), Japan (2.3%), Chile (1.7%), Spain (1.4%), Thailand (1.4%), USA (1.1%), France (1.1%), and Italy (0.7%) (Figure 3). The major aquaculture species or groups differ by country (Table 1).
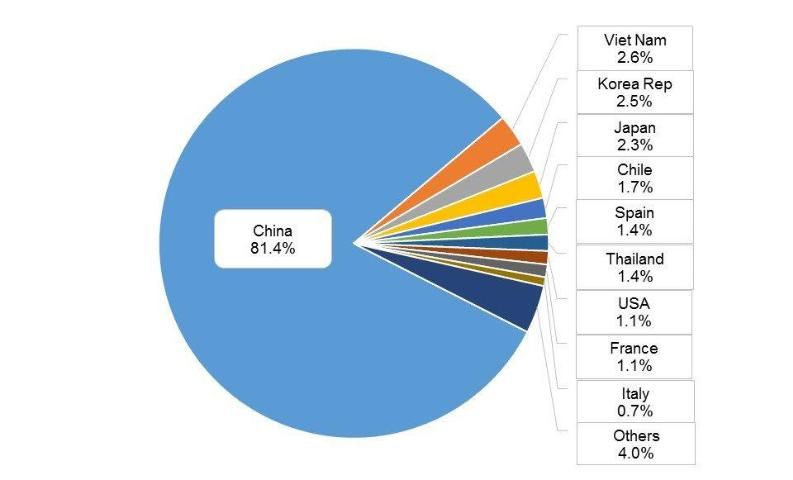
Credit: FAO 2012
Molluscan Shellfish Aquaculture in the United States
In the United States, molluscan aquaculture is a $329 million industry involving 756 farms in 18 states (USDA 2014). Nationwide, the top three species in aquaculture sales in 2013 were the Pacific oyster Crassostrea gigas ($87million), the eastern oyster Crassostrea virginica ($68 million), and the hard clam Mercenaria mercenaria ($65 million) (Figure 4). The Pacific oyster is cultured on the west coast of the United States in the states of Alaska, Washington, Oregon, and California. The eastern oyster is cultured on the east coast of the United States and in the Gulf of Mexico in the states of Maine, Massachusetts, New York, Connecticut, Rhode Island, New Jersey, Maryland, Virginia, North Carolina, South Carolina, Georgia, Florida, Alabama, and Louisiana. The hard clam is also cultured on the east coast in the states of Massachusetts, New York, Connecticut, Rhode Island, New Jersey, Maryland, Virginia, North Carolina, South Carolina, Georgia, and Florida.
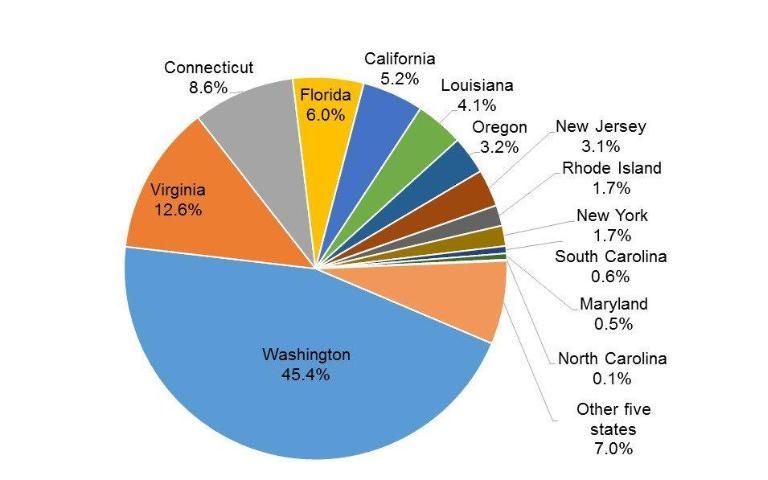
Among the 18 states with molluscan aquaculture, the state of Washington has the greatest aquaculture sales, at $149.3 million, or 45.4% of the total US sales. The molluscan aquaculture industry in the state of Washington is also the most diversified and includes Pacific oysters, mussels, manila clams, geoducks, and other molluscs. The states of Virginia ($41.5 million), Connecticut ($28.3 million), Florida ($19.6 million), and California ($17.0 million) trail Washington in molluscan aquaculture sales (Figure 5). In Virginia and Connecticut, the major species are eastern oysters and hard clams; in Florida, the major aquaculture species is the hard clam with some production of eastern oysters and sunray venus clams; and, in California, the aquaculture species are Pacific oysters, abalones, and clams (USDA 2014).
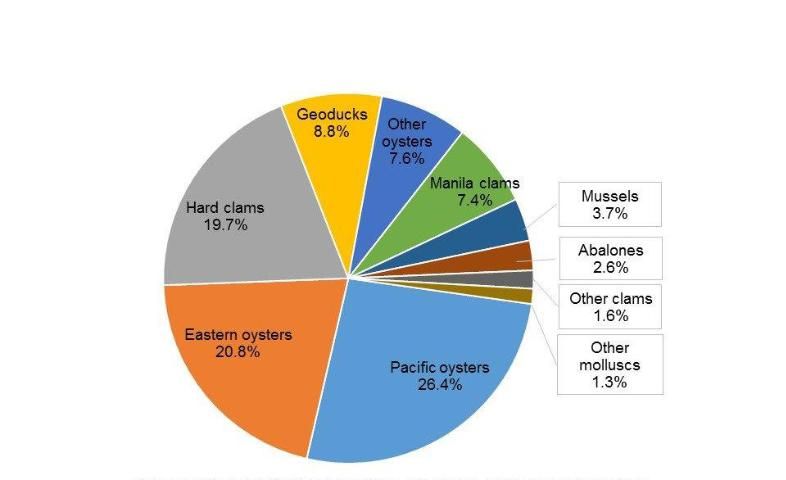
Credit: USDA 2014
Credit: USDA 2014
Molluscan Shellfish Habitats and Aquaculture
Most cultured molluscan species are bivalves; only a few gastropods, such as abalones, are cultured. Feeding and reproductive modes may account for the relative ease of culturing bivalves over gastropods. Bivalve molluscs are typically filter feeders of phytoplankton and are primarily free spawning with external fertilization. Due to the very large and diversified taxonomic class of gastropods, their reproduction, feeding, and habitat types vary greatly. Gastropod molluscs may be herbivores, carnivores, parasites, or omnivores, and exhibit complex mating behaviors (courtship) with either internal or external fertilization. Abalones are the primary gastropod species for aquaculture. Like bivalves, they are free spawning, with external fertilization, and have a free-swimming larval stage that feeds on phytoplankton. Following metamorphosis (a morphological transformation from swimming larvae to juvenile) the herbivorous spat (juveniles after metamorphosis) graze on benthic encrusting algae or bacterial films using their radula (an anatomical structure used in feeding that is unique to molluscs and present in all of them except bivalves). As abalone grow, they increasingly rely on macro algae as food sources; aquaculture of abalones requires providing microalgae-coated plates during larval metamorphosis and seaweed during growout (the culture period from seed to market-sized adults).
In general, bivalves for aquaculture typically have one of three habitat lifestyles: 1) endobenthic burrowing (bivalves that live within the sediments), 2) epibenthic cemented (bivalves that live on the surface of sediments), or 3) epibenthic free-living (non-attached) or attached with byssal thread (a keratin and protein filament secreted from the muscular foot). Most clams have an endobenthic burrowing lifestyle. The muscular, hatchet-shaped foot and round or elongated shell shape of most species of clams aid them in burrowing. Depending on the species, the preferred benthic substrate may be sand, mud, or a mixture, and burrowing depth may be from centimeters to meters. Successful aquaculture of these species requires suitable substrates for growout in open water environments. Oysters exhibit an epibenthic cemented lifestyle. When ready to metamorphose, free-swimming oyster larvae seek a suitable hard substrate, such as calcareous shell, and cement themselves down with a glue-like protein; they will remain attached for life and lose their foot after metamorphosis. Oysters can close tightly, providing protection from predators, desiccation, and other harsh environmental conditions. Culture of oyster species requires consideration of suitable substrate types for metamorphosis. During growout, oysters are cultured in a variety of systems, which vary from on-bottom to off-bottom, suspended or floating, with or without predator protection. Air drying and other control methods must be used to prevent biofouling of culture gear and oysters. Scallops and mussels have an epibenthic free or byssal attached lifestyle. They are capable of abandoning their byssal threads temporarily and swimming to avoid predators or to move to a more suitable location. Scallops usually have two light-weight and curved symmetric valves, shell ears (the extended part of shell near the hinge) with a byssus orifice, and a ventrally flattened shape to enable them to create a directed water flow and ensure a firm contact with the substrate. Culture of scallop species requires providing suitable substrates for larval metamorphosis in the hatchery; commonly used substrates include window screen, polyethylene threads, or coir (natural fibre from coconut or palm tree) threads/ropes. Floating or suspended cages are used for scallop growout. Mussels can strongly attach to substrates by many byssal threads. Most mussels have two symmetric and thin shells hinged together tightly. Culture of mussel species also requires suitable substrates for larval metamorphosis in the hatchery. Growout culture usually occurs on ropes in floating rafts or off-bottom poles without cages due to their strong byssal attachment.
Molluscan Shellfish Aquaculture Stages and Methodologies
Molluscan shellfish aquaculture includes three biological or cultural stages: production of seed, juvenile nursery culture, and growout of sub-adults to harvest size. The aquaculture methods, culture gear, and routine maintenance must be based on a thorough understanding of the biological characteristics of the target species.
Seed Production
Two approaches are currently employed for seed production of molluscs: hatchery production and seed collection (collecting seed from natural resources by providing suitable substrates). In hatchery production, seed are produced under controlled conditions (FAO 2004). Methods include broodstock selection and conditioning (to accelerate sexual maturity), spawning and gamete collection, fertilization, swimming larval culture, metamorphosis, and post-metamorphosis rearing. The water for hatcheries is usually treated by sedimentation, filtration, sterilization or disinfection to meet the needs of both the larvae and their multi-species microalgal food. Hatcheries have the advantage of increasing the quantity and quality of seed production, producing seed out of the natural spawning season, and providing opportunities for broodstock breeding and various genetic manipulations. In addition, hatchery-produced oyster pediveliger larvae (i.e., pre-settlement larvae) can be shipped to other locations for setting and further nursing, which is called remote setting. If allowed to settle on shell (i.e., cultch), the resulting seed, referred to as "spat-on-shell," may be used in extensive oyster aquaculture efforts, as well as restoration efforts. This method is in contrast to single oyster seed production, referred to as cultchless, which results in intensive culture of individual oysters in various containers (e.g., bags, cages).
The practice of collecting seed from the wild is usually performed by providing suitable substrates for metamorphosing larvae, such as rock piles, bamboo poles, and shell strings, or by tilling and cleaning natural substrates. Site and substrate selection, as well as timing, is critical for obtaining seed and minimizing labor cost. These methods have been used for oyster, mussel, and clam aquaculture for over two thousand years. Placing shell substrate, or cultch, during the natural spawning season is still a viable method of collecting oyster seed and supports extensive aquaculture efforts in many coastal states.
Nursery
The nursery stage is an intermediate stage between the hatchery and growout aimed to increase seed size in a protected environment prior to out-planting, thereby improving survival and growth. Based on the habitat requirements of the aquaculture species, nursery methods may include land-based flow-through systems (up- or down-wellers, raceways) (FAO 2004), floating or submerged nets or trays in intertidal ponds with enhanced microalgae culture, floating or bottom nets, or trays in open waters. Optimal seed density in the nursery varies depending on the species, seed size, food availability, and water flow. Routine maintenance during the nursery stage includes cleaning the culture systems, measuring water quality, and monitoring for disease.
Growout
After the nursery stage, larger seed or juveniles are out-planted and grown to harvest size during the growout stage of aquaculture. The first step is site selection; growout may take place in open waters, offshore, in coastal ponds, or in intertidal areas. All growout operations, whether located on private land or on public lands leased from a government entity must follow the federal and state regulations addressing shellfish harvesting water classifications, environmental issues, use of waterways, public health, and food safety, etc. In addition, understanding of the culture environmental conditions, such as wave and tidal height, water depth, substrate, salinity, temperature, phytoplankton biomass, and predator species and abundance, is the basis for culture method selection, layout, and management. Growout culture methods are species-specific and include on-bottom, off-bottom poles or racks, floating rafts or longline systems, and cages or nets of various designs. The selection of growout culture gear must consider the biological characteristics of the cultured species, the physical, chemical and biological characteristics of the culture environment, and the ease of routine maintenance and harvest.
Summary
Overall, molluscan shellfish aquaculture provides high quality and high value seafood for human consumption. In addition, filter-feeding molluscan shellfish provide environmentally beneficial ecosystem services, such as nutrient extraction and water filtration, to the environment in which they are grown. Shellfish aquaculture is the most environmentally sustainable culture form, and will continue to expand with the development of new technologies and improved production practices.
Annotation
Most data used in this publication were obtained from the database of the Fisheries and Aquaculture Department of the Food and Agricultural Organization (FAO) (www.fao.org/fishery/statistics/collections/en) and the Census of Aquaculture in the United States Department of Agriculture (USDA, http://www.agcensus.usda.gov/Publications/Census_of_Aquaculture/). The fisheries and aquaculture database within FAO is structured on a global and regional basis with comprehensive statistics, and is issued annually with updated data collection. The USDA census is conducted and published every five years. Readers are encouraged to visit these two websites for any updated information about molluscan shellfish aquaculture production.
References Cited
Delgado, C. L., N. Wada, M. W. Rosegrant, S. Merjer, and M. Ahmed. 2003. Outlook for Fish to 2020: Meeting Global Demand. International Food Policy Research Institute and World Fish Center. Washington D. C., USA.
Diana, J. S. 2009. Aquaculture Production and Biodiversity Conservation. Bioscience 59, 27–38.
FAO. 2004. Hatchery Culture of Bivalves: A Practical Manual. In: FAO Fisheries Technical Paper 471.
FAO. 2012. Year book, Fishery and Aquaculture Statistics. URL http://www.fao.org/fishery/statistics/programme/publications/all/en.
FAO. 2014. The State of World Fisheries and Aquaculture Opportunities and Challenges. p. 243, Rome.
USDA. 2014. 2012 Census of Agriculture: Census of Aquaculture (2013), released on September, 2014. p. 98.