Introduction
Hemicheyletia wellsina (De Leon) (Figure 1) is a predatory mite in the family Cheyletidae. The life history of species in this family is very diverse. About 75% of cheyletid mites are predators of other arthropods and the rest are parasitic on birds and mammals (Bochkov and O'Connor 2004, Krantz and Walter 2009). Hemicheyletia wellsina was recently discovered in an unsprayed greenhouse at the University of Florida, Gainesville, living on Phalaenopsis and Dendrobium orchids, and assumed to be feeding on orchid pests such as spider mites, tenuipalpid mites, and mealybugs that were present on the orchids. Because there was no published information on the biology or effectiveness of this species as a natural enemy of orchid pests, colonies were initiated in the Entomology and Nematology department at the University of Florida to study the biology of Hemicheyletia wellsina. A better understanding of this predator's biology could have implications for hobbyist orchid growers. Colonies of Hemicheyletia wellsina were maintained on two-spotted spider mite Tetranychus urticae (Koch) prey. This predator fed on all active stages but did not feed on eggs.
Credit: Haleigh Ray, UF/IFAS
Distribution
Hemicheyletia wellsina was originally described in Trinidad, and is currently distributed through Malaysia, Taiwan, the Philippines, and north and central Africa (Fain et al. 2002). This species has been reported in many counties in southern Florida, and as far north as Alachua County, Florida.
Description
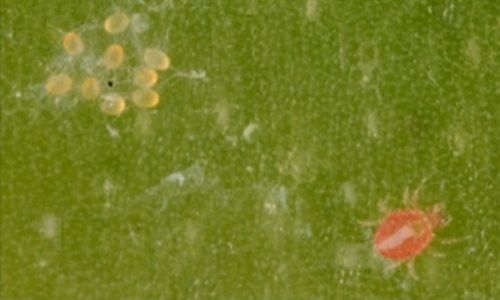
Hemicheyletia wellsina is a small, round mite (Figure 2). The larvae measure approximately 0.12 mm in width and 0.15 mm in length. Adults average approximately 0.25 mm in width and 0.29 mm in length, with adult females being about 20% larger than adult males. Like all cheyletids, they have short, stylet-like mouthparts called chelicerae and large pincer-like palps for grasping prey (Baker and Wharton 1952; Volgin 1987; Ray and Hoy 2014a) (Figure 2). When the larvae hatch they have six legs but, after molting, both nymphs and adults have eight legs. Hemicheyletia wellsina becomes a brighter shade of red with each molt, and sometimes adults are a deep red color (Figure 3).

Credit: Haleigh Ray, UF/IFAS

Credit: Haleigh Ray, UF/IFAS
Life Cycle and Behavior
After hatching from the egg, Hemicheyletia wellsina has four life stages: the larvae, a first nymphal stage, a second nymphal stage, and adult males and females. In most mite species, the first and second nymphal stages are called protonymph and deutonymph, respectively. Because a third nymphal state (tritonymph) occurs in some species of this family, the nymphal stages are simply referred to as first and second here. Hemicheyletia wellsina lays eggs in groups, or clutches, that are covered by fine silk webbing (Figure 4). This webbing probably provides both protection from predators and helps to regulate the relative humidity that the eggs experience (Ray and Hoy 2014c). When larvae first hatch they do not survive if removed from under the silk. After about 3 days, the larvae could be moved to a clean leaf disc and survive. As soon as the Hemicheyletia wellsina larvae move away from the silk, they could then feed on much larger prey.
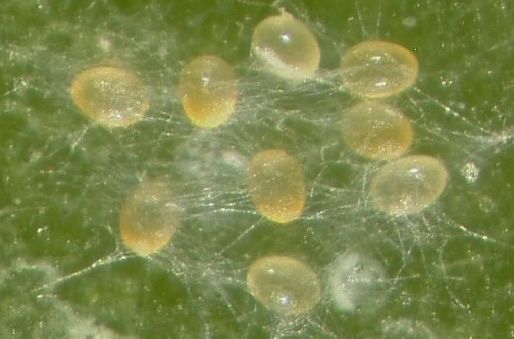
Credit: Haleigh Ray, UF/IFAS
The average lifespan of Hemicheyletia wellsina from hatching until death is approximately 30 days, with individuals living as few as 16 days and as long as 47 days at 25°C with a relative humidity of 45–55% and a 16 h light and 8 h dark photoperiod. The time between when the eggs are laid until they hatch is three to five days, and seven to 10 more days are required to reach adulthood. The adult females lay two to three clutches of silk-covered eggs, usually with seven to eight eggs per clutch. Each adult female deposits an average of 18.5 eggs throughout her lifetime (Ray and Hoy 2014a).
Hemicheyletia wellsina adult females were observed to feed on Phalaenopsis mites (Tenuipalpus pacificus Baker), two-spotted spider mites (Tetranychus urticae (Koch)), one life stage of Boisduval scale (Diaspis boisduvalii Signoret), and the western orchard predatory mite (Metaseiulus occidentalis (Nesbitt)) (Figures 5–8).

Credit: Haleigh Ray, UF/IFAS

Credit: Haleigh Ray, UF/IFAS

Credit: Haleigh Ray, UF/IFAS

Credit: Haleigh Ray, UF/IFAS
Hemicheyletia wellsina typically captures and feeds on these species by grasping the leg of the prey as it comes in contact with the palps of Hemicheyletia wellsina. Like many cheyletids, Hemicheyletia wellsina uses the 'ambush' method of prey capture described by Wharton and Arlian (1972). Hemicheyletia wellsina remains stationary on the leaf surface with its palps opened wide. When an arthropod comes into contact with the palps, they rapidly close, trapping the prey. Within seconds the prey becomes immobile, which suggests that Hemicheyletia wellsina injects a toxin in to the prey as do several cheyletid species (Gerson et al. 2003). This allows them to feed on prey that is much larger, and even larval Hemicheyletia wellsina can feed on adults of the larger predatory phytoseiid Metaseiulus occidentalis (Figure 9).

Credit: Haleigh Ray, UF/IFAS
Adult Hemicheyletia wellsina females were individually placed on a leaf with the adult females or larvae of the predatory mite Metaseiulus occidentalis. Every time the predators came in contact with each other, Hemicheyletia wellsina captured and fed on Metaseiulus occidentalis (Figure 8). However, without access to food, a newly emerged adult female Hemicheyletia wellsina can live for approximately 10 days without feeding (Ray and Hoy 2014a). Individuals do not require many prey to survive, with adult Hemicheyletia wellsina females feeding on an average of 1.5 adult spider mites per day, and an average of 27 adult spider mites throughout their lifetime.
Habitat
Hemicheyletia wellsina has been found on native wild plants and flowers as well as in leaf litter, flowers of coconut (Cocos nucifera), Hibiscus sp., asparagus fern (Asparagus plumosus), Philippine tea tree (Ehretia microphylla), yellow root (Arcangelisia flava), Philippine fishtail palm (Caryota cumingii), bitter vine (Mikania micrantha), buffalo grass (Paspalum conjugatum), butterfly-pea (Clitorea ternatea), chestnut (Castanea crenata), Melaleuca leucodendron, Wedelia biflora, and wild blackberry (Rubus ulmifolius) (Tseng 1972; Corpuz-Raros and Sotto 1977; Ehara and Ibrahim 1988; Fain et al. 2002). Although Hemicheyletia wellsina has been found mostly on wild plants, it was collected from Phalaenopsis and Dendrobium orchids in a greenhouse. No records indicate that Hemicheyletia wellsina occurs in agricultural systems.
Economic Importance
Orchids are susceptible to many types of arthropod pests. Scales, mealybugs, thrips, mites, aphids, and whiteflies are most common (Bottom 2012). Of these, the most difficult to control are mites, which require a high rate of pesticide use on ornamental plants (Naher et al. 2005). Hemicheyletia wellsina can feed on two species of plant-feeding mite (Tetranychus urticae and Tenuipalpus pacificus) and one species of scale (Diaspis boisduvalii), but may not feed on a sufficiently large number of individuals to alone prevent damage to the orchids especially if pest densities are high. In a study by Ray and Hoy (2014b), two reduced-risk pesticides were tested on Hemicheyletia wellsina to determine if they could be used in addition to Hemicheyletia wellsina for control of orchid pests. Grandevo (Marrone Bio Innovations 2012) and a combination of Silwet L-77 and 2% horticultural oil were sprayed directly on Hemicheyletia wellsina adults (Mention of these products does not endorse their use). Grandevo resulted in low mortality of Hemicheyletia wellsina, while Silwet L-77 and horticultural oil caused a high rate of mortality to Hemicheyletia wellsina (Ray and Hoy 2014b).
Ideally, predators used for biological control have a high prey searching ability, prey on a specific pest species, can increase in numbers more rapidly than the targeted prey, and have the ability to adapt to new environments (Rosen and Huffaker 1983). Hemicheyletia wellsina does not appear likely to be an effective natural enemy in agricultural crops as an introduced predator. This predator is unlikely to be able to suppress spider mites at a sustainable level due to its low reproductive rate, longer developmental time, and low feeding rate. However, Hemicheyletia wellsina could be beneficial in natural biological control in natural ecosystems, where pest densities are lower. If Hemicheyletia wellsina is present, it could limit pests and reduce plant damage in natural ecosystems or in landscape settings, although this needs to be confirmed. Hemicheyletia wellsina has been observed to feed on Phalenopsis mites (Tenuipalpus pacificus), two-spotted spider mite (Tetranychus urticae), and one life stage of Boisduval scale (Diaspis boisduvalii).
Acknowledgements
We thank Dr. Cal Welbourn of the Division of Plant Industry, Gainesville FL and Dr. Andre Bochkov of the Department of Parasitology, Zoological Institute of the Russian Academy of Sciences for species identification. We also thank Godfrey Maina for help with colony maintenance. We thank Lyle Buss of the UF/IFAS Department of Entomology and Nematology for assistance with photographs, and Dr. Howard Frank for review of this article. This project was supported by the Davies, Fischer and Eckes Endowment in biological control to Marjorie A. Hoy.
Selected References
Baker EW, Wharton GW. 1952. An introduction to acarology. The Macmillan Company, NY, pp 232–235.
Bochkov AV, OConnor BM. 2004. Phylogeny, taxonomy and biology of mites of the genera Chelacheles and Neochelacheles (Acari: Cheyletidae). Invertebrate Systematics 18: 547–592.
Bottom S 2012. St. Augustine Orchid Society: Orchid Pests and Diseases. http://staugorchidsociety.org/culturepests-pests.htm. Accessed 6 February 2013.
Corpuz-Raros LA, Sotto JM. 1977. Systematic studies of Philippine cheyletid mites (Acarina: Cheyletidae). II. New Species and new records. Kalikasan, Philippine Journal of Biology 6: 143–170.
Ehara S, Ibrahim AG. 1988. Cheyletid mites associated with plants in the Malay Peninsula, with description of a new species (Acarina: Cheyletidae). Proceedings of the Japan Academy, Series B, Physical and Biological Sciences 64: 237–240.
Fain A, Bochkov AV, Corpuz-Raros LA. 2002. A revision of the Hemicheyletia generic group (Acari: Cheyletidae). Entomologie 72: 27–66.
Gerson U, Smiley RL, Ochoa R. 2003. Mites (Acari) for pest control. Blackwell Publishing, Oxford, UK, pp 103–111.
Krantz GW, Walter DE. 2009. A Manual of Acarology, Third Edition. Texas Tech University Press, Lubbock, TX, pp 57–304.
Marrone Bio Innovations. (2012). Grandevo. http://www.marronebioinnovations.com/products/brand/grandevo/. Accessed 12 September 2014.
Naher N, Islam W, Haque MM. 2005. Predation of three predators on two-spotted spider mite, Tetranychus urticae Koch (Acari: Tetranychidae). Journal of Life and Earth Sciences 1: 1–4.
Ray HA, Hoy MA. 2014a. Evaluation of the predacious mite Hemicheyletia wellsina (Acari: Cheyletidae) as a predator of arthropod pests of orchids. Experimental and Applied Acarology, DOI 10.1007/s10493-014-9833-8.
Ray HA, Hoy MA. 2014b. Effects of reduced-risk insecticides on three orchid pests and two predacious natural enemies. Florida Entomologist 97: 972–978.
Ray HA, Hoy MA. 2014c. Role of silk webbing in the biology of Hemicheyletia wellsina (Acari: Cheyletidae). International Journal of Acarology In press.
Rosen D, Huffaker CB. 1983. An overview of desired attributes of effective biological control agents, with particular emphasis on mites. In: Hoy MA, Cunningham GL, Knutson L (eds) Biological Control of Pests by Mites. University of California. Berkeley, California. pp 2–11.
Tseng YH. 1972. A new cheyletid mite Hemicheyletia arecana n. sp. from Taiwan (Acarina: Prostigmata). Plant Protection Bulletin 14: 8–10.
Volgin VI. 1987. Acarina of the family Cheyletidae of the world. Amerind Publishing Co. Pvt. Ltd., New Delhi, India, pp 3–39.
Wharton GW, Arlian LG. 1972. Predatory behavior of the mite Cheyletus aversor. Animal Behaviour 20: 719–723.