Summary
Water from rainfall or irrigation can collect in the central tanks or axils of bromeliads planted as ornamentals in subtropical and tropical gardens. The aquatic fauna that occupy these small pools in bromeliad axils and tanks often include the immature stages of mosquitoes. In Florida, mosquitoes that develop in this microhabitat include native species of Wyeomyia as well as the invasive container-generalist species Aedes albopictus and Aedes aegypti, which may transmit mosquito-borne viruses such as dengue and Zika to humans. Experiments conducted under semi-natural conditions in Florida using the popular ornamental bromeliad Billbergia pyramidalis showed that small larvae of A. albopictus were unable to complete development in the presence of large larvae of Wyeomyia spp., leading to the conclusion that competition with the non-vector native species provides a biological control of the dangerous, invasive vector mosquito.
In 2016 during a Zika virus outbreak in Miami, ornamental bromeliads were removed from public gardens in the belief that they contributed to production of the vector A. aegypti. Similarly, in 2008 during a dengue epidemic in Rio de Janeiro, municipal mosquito control workers extirpated bromeliads from private gardens to eliminate supposed sources of A. aegypti. Subsequent surveillance and identification of mosquitoes extracted from bromeliad tanks in an urban botanical garden in Rio revealed the presence of very few immatures of the vector species A. aegypti and A. albopictus, and currently there is no validation for the elimination of bromeliads during outbreaks of mosquito-borne viruses.
However, even in the absence of vector-borne diseases, water-holding bromeliads may be sources of nuisance biting mosquitoes like Wyeomyia spp. This publication provides recommendations for environmentally acceptable ways to control the production of nuisance mosquitoes in bromeliads, suggestions for planting species of bromeliads less likely to favor the production of mosquito-borne disease vectors, and general advice about personal protection against mosquito bites.
Introduction
Due to their attractive foliage and flowers and ease of propagation, exotic tank bromeliads are popular ornamental landscaping plants in areas like Florida with tropical and subtropical climates (Figure 1; Frank and Lounibos 2009). Unfortunately, tank bromeliads that hold water also frequently produce biting mosquitoes. Whether the mosquitoes produced by ornamental bromeliads are responsible for transmitting viruses to humans has been a source of international controversies during mosquito-borne virus outbreaks. This publication is intended for all audiences and provides background information on mosquitoes, bromeliads, their relationships, and the controversies that have taken place surrounding them. Recommendations for reducing the number of mosquitoes produced by bromeliads are also provided.

Credit: E. A. Buckner and A. L. Romero-Weaver, UF/IFAS
Mosquitoes
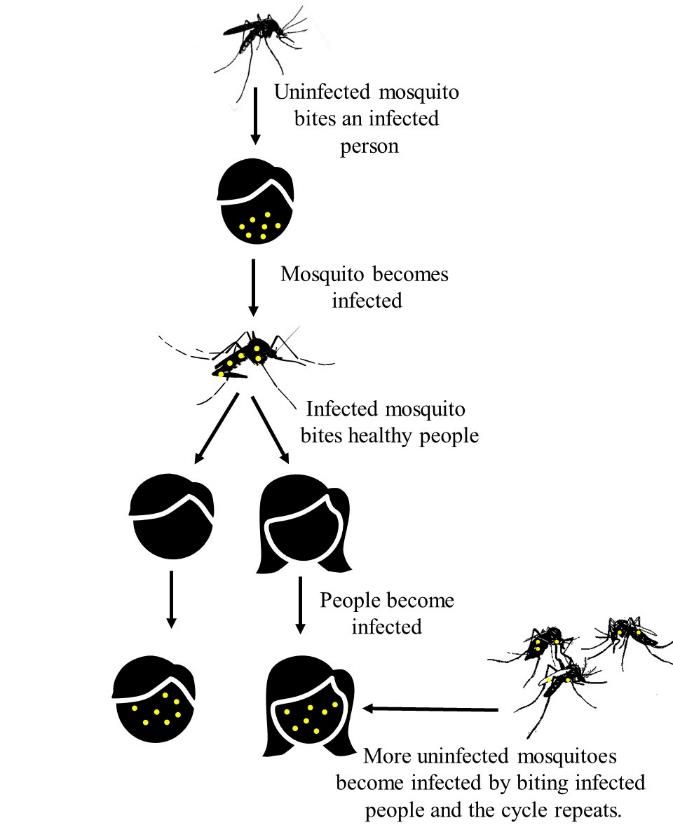
There are more than 3500 species of mosquitoes worldwide, which can be found on all continents except Antarctica. All mosquitoes possess a life cycle made up of four stages: egg, larva, pupa, and adult (Figure 2). Mosquito eggs are laid singly or in rafts on the surface of water or on moist substrate. All mosquito eggs need water to hatch. The larval and pupal life stages are aquatic. Most mosquito larvae obtain nutrients by filter feeding on microorganisms and organic debris in the water of their aquatic habitats. As mosquito larvae grow, they pass through a total of four increasingly larger larval instars (stages). Once a mosquito larva has obtained all the nutrition it needs, it transforms into a pupa, which is the mosquito equivalent of a butterfly’s chrysalis. Pupae do not feed but can swim to avoid predation. Adult mosquito characteristics form during the pupal stage, which usually lasts 2 days. After the transformation is complete, a fully formed adult mosquito emerges from a slit in on the dorsal side of the pupa, leaving its pupal skin behind. The new adult mosquito rests on the surface of the water briefly, and then flies away to begin the terrestrial phase of its life cycle. Both adult male and female mosquitoes feed on plant nectar as a source of carbohydrates for energy. Typically, after an adult female mosquito has mated with a male, she will bite and take one or more blood meals to finish egg development. If infected with a pathogen like a virus, protozoan, or nematode roundworm, the mosquito may transmit the pathogen to the host while blood-feeding (Figure 3).
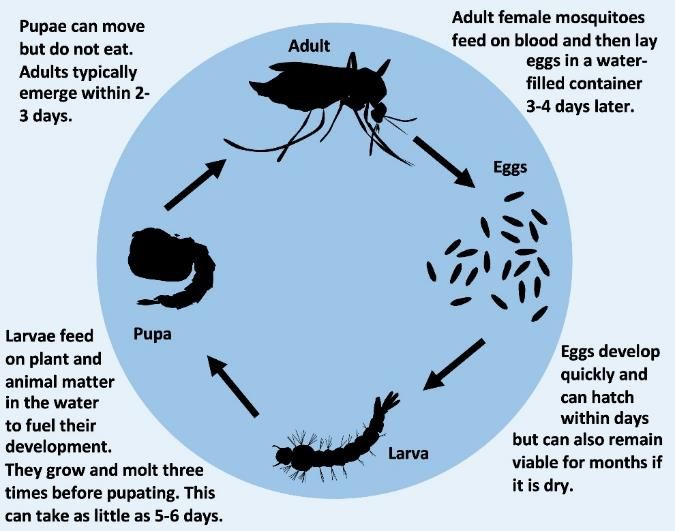
Credit: E. P. Caragata, UF/IFAS
Credit: A. L. Romero-Weaver, UF/IFAS
Bromeliads
Bromeliads are perennial flowering plants of the pineapple family Bromeliaceae. There are over 3,000 bromeliad species that are native primarily to the subtropical and tropical Americas (Benzing 1990, Holst and Luther 2004). Sixteen species are native to the state of Florida (Luther and Benzing 2009). For more information on Florida’s native bromeliads, see https://edis.ifas.ufl.edu/publication/uw205. While a few exotic bromeliads like pineapples are grown as crops, most are grown in tropical and subtropical climates for use as ornamental landscaping plants due to their attractive foliage and flowers (Figure 1), inexpensiveness, and low maintenance (Frank and Lounibos 2009, Frank 2021). Bromeliads can also be grown in greenhouses or indoors in temperate climates (Figure 4).

Credit: UF/IFAS
Many bromeliads are described as epiphytic, which means that they grow on other plants (Benzing 1990; Figure 5). The roots of epiphytic bromeliad plants help to anchor them to the plants or the surfaces that they grow on (Benzing 2000). Epiphytic bromeliads use trichomes (specialized hairs that cover their leaves) to absorb water and nutrients (Luther and Benzing 2009, North et al. 2013). Additionally, in most bromeliads, intricately arrayed leaves form a spiral rosette with a tank-like structure at its center where rainwater and debris can collect (Figure 6). Water can also collect at the axil (base) of each bromeliad leaf (Ladino et al. 2019; Figure 7). The water-filled tanks and axils can trap pollen, dead leaves, twigs, and seeds falling from trees, which break down to form a nutritive soup available to the bromeliads and other organisms inhabiting the bromeliads like mosquito larvae.

Credit: L. P. Lounibos, UF/IFAS

Credit: L. P. Lounibos, UF/IFAS

Credit: Durland Fish, Yale University
Can bromeliads produce mosquitoes?
Yes. Water that collects in a bromeliad tank or at the axils of bromeliad leaves is an example of a phytotelm, a small water body held by terrestrial plants, which can be used by container-inhabiting mosquitoes for oviposition (egg laying) and pre-adult development (Figures 8 and 9). Container-inhabiting mosquitoes are called such because of the typically small aquatic habitats that they occupy during their immature life stages. Container-inhabiting mosquitoes may be subdivided into generalist species, such as the invasive yellow fever mosquito (Aedes aegypti) and Asian tiger mosquito (Aedes albopictus), whose adult females can transmit chikungunya, dengue, and Zika viruses to humans, or phytotelm specialists, such as the Florida native bromeliad-inhabiting mosquito species Wyeomyia vanduzeei and W. mitchellii, whose larvae are typically found only in the axils of bromeliads (Kitching 2000, Frank and Lounibos 2008). For more information on Florida container mosquitoes, please see https://edis.ifas.ufl.edu/publication/IN1315.

Credit: J. Newman, UF/IFAS

Credit: L. E. Reeves, UF/IFAS
Do all bromeliads produce mosquitoes that can transmit viruses to humans?
No. Many factors including a bromeliad’s location, species, and size can influence the mosquito species found within it (O’Meara et al. 2003). Raban (2006) found that more Asian tiger mosquito eggs were laid in tanks of Neoregalia cruenta, an exotic bromeliad, in paired comparisons with similarly sized Billbergia pyramidalis, another tank bromeliad popularly grown in Florida. Also, as mentioned earlier, the mosquito species W. mitchellii and W. vanduzeei are phytotelm specialists, and their larvae are typically found only in bromeliads. If abundant, Wyeomyia mosquitoes can be a biting nuisance to bromeliad growers and their neighbors, but they are not known to transmit viruses that cause diseases in humans (Edman and Haeger 1978). In fact, the presence of Wyeomyia mosquito larvae in bromeliads may actually reduce the number of yellow fever and Asian tiger mosquito larvae developing in them. Multiple surveys of the mosquito fauna in southern Florida demonstrated that immature stages of the yellow fever mosquito or the Asian tiger mosquito were relatively uncommon if Wyeomyia mosquito larvae were abundant in the same bromeliad tanks or axils (Frank et al. 1988, Lounibos et al. 2003). For example, Frank et al. (1988) surveyed mosquito larvae in the exotic bromeliad Billbergia pyramidalis in four southern Florida cities and found approximately 20,000 immature Wyeomyia mosquito larvae but less than 200 immature yellow fever mosquitoes. Conversely, Lounibos et al. (2003) documented a dramatic increase in Asian tiger mosquito abundance in exotic bromeliads north of Orlando in central Florida where W. vanduzeei and W. mitchellii are rare due to the scarcity of their native bromeliad host plant Tillandsia utriculata. This inverse relationship between Wyeomyia and Asian tiger mosquito larval abundances observed in bromeliads along a latitudinal gradient in Florida suggests that Wyeomyia may inhibit the production of Asian tiger mosquitoes in exotic bromeliads (Lounibos et al. 2003).
Also, Lounibos et al. (2003) conducted experiments in tanks of B. pyramidalis bromeliads set in semi-natural outdoor enclosures to determine whether mature larvae of Wyeomyia spp. (a) inhibited oviposition (egg-laying) by gravid (pregnant) Asian tiger mosquitoes or (b) impeded growth and development of early-stage larvae of Asian tiger mosquitoes. Results clearly showed that Wyeomyia spp. larvae did not inhibit oviposition by Asian tiger mosquitoes in bromeliads, but the large larvae of the native phytotelm specialists did inhibit survivorship and development of early-stage Asian tiger mosquito larvae. Thus, larval competition between native Wyeomyia mosquitoes and the invasive Asian tiger mosquito is thought to be the mechanism responsible for the inverse relationship in abundances of these mosquitoes in samples from bromeliads in south and central Florida (Lounibos et al. 2003).
Controversies over mosquitoes and bromeliads during mosquito-borne virus outbreaks
In Rio de Janeiro, Brazil, dengue is an endemic and regular public health threat, especially during the rainy season. During a particularly fierce dengue epidemic in 2008, mosquito control authorities in Rio began to destroy exotic bromeliads in the gardens of residents, based on the untested hypothesis that yellow fever mosquitoes, which transmit dengue virus to humans, were commonly produced in the tanks (Mocellin et al. 2009). To fill a research gap on the relative importance of terrestrial bromeliads for producing yellow fever mosquitoes or Asian tiger mosquitoes in urban Rio, Mocellin et al. (2009) sampled the immature stages of mosquitoes monthly for one year from the tanks and axils of ten bromeliad plants located in Rio’s Jardim Botanico, an old public garden located in relict Atlantic coastal rain forest bordered by favelas (slums) with regular dengue virus transmission. Among 2,816 immature mosquitoes identified from these samples, only two yellow fever mosquitoes and 5 Asian tiger mosquitoes were detected. The majority of mosquito species identified were Culex (Microculex) and Culex (Culex), native, non-vector specialists of Brazilian bromeliads that might play similar roles as Wyeomyia spp. in Florida to minimize the production of invasive Aedes spp. generalists from these phytotelmata (small water bodies held by terrestrial plants). A plaque commemorating these research findings was subsequently erected in the Jardim Botanico (Figure 10). Additionally, working north of Rio in Vitoria, Santos et al. (2011) concluded from mosquito surveillance that bromeliads growing naturally on rocky outcrops near houses with known dengue transmission did not contribute to yellow fever mosquito production.

Credit: L. P. Lounibos, UF/IFAS
Based on the concern that exotic bromeliads planted as ornamentals in public spaces in the city of Miami were producing yellow fever mosquitoes during the Zika outbreak of 2016, Miami-Dade Mosquito Control authorized the removal of hundreds of bromeliads (Gomez 2016). It remains controversial whether this action, undertaken to protect public health, was warranted. Elsewhere in Miami, subsequent surveillance of exotic bromeliad phytotelmata after the outbreak indicated that the yellow fever mosquito was the most abundant species detected, being slightly more common than Wyeomyia spp. (Wilke et al. 2018). In other places within Florida, recent surveillance of mosquito abundances in tanks and axils of exotic bromeliads showed that the Asian tiger mosquito was the most prevalent species in samples in urban St. Augustine, followed by W. vanduzeeii (Xue et al. 2018). Unfortunately, larval densities, pupal numbers, and bromeliad species, all important for management decisions based on the data, were not provided in these surveys.
Currently, data are insufficient to support removal or destruction of tank bromeliads as a means of vector control directed at invasive yellow fever or Asian tiger mosquitoes. Before remedial actions are applied to ornamental bromeliads that form phytotelmata, researchers or vector control workers need to first establish that major numbers of these Aedes spp. vectors are developing to the pupal stage in these plants. Because native Wyeomyia spp. mosquitoes, which do not transmit pathogens to humans, exert a natural biocontrol effect against yellow fever and Asian tiger mosquitoes, maintenance of these species in bromeliad tanks and axils should be encouraged. More experimental research is needed to define the conditions under which and mechanisms whereby Wyeomyia spp. specialists suppress Aedes spp. generalists in these container habitats.
Recommendations for Mosquito Control in Bromeliads
First and foremost, understand that bromeliads require ongoing maintenance to prevent production of mosquitoes! Though Wyeomyia spp. mosquitoes may not transmit harmful viruses as Asian tiger and yellow fever mosquitoes can, they can bite humans and become a nuisance if in high numbers. Therefore, if you are unable to provide the ongoing maintenance necessary to prevent bromeliads from producing potential nuisance-causing mosquitoes, consider planting other plants with attractive foliage in your landscape.
However, if you are a bromeliad enthusiast and your landscape is made up of many tank-forming bromeliads, you can avoid the rapid production of mosquitoes in your backyard using one or a combination of these management recommendations:
- Flush out eggs, larvae and debris accumulated in bromeliad axils and tanks using a strong jet of water from a hose at least once a week.
- Add larvicides to the water accumulated in the leaves of bromeliads. There are two commercially available larvicide active ingredients, methoprene and Bacillus thuringiensis subspecies israelensis (commonly known as Bti), that can be found in most home and garden supply stores. When applied correctly using instructions found on product labels, these larvicides do not harm plants, people, pets, or the environment (CDC 2020). They are usually sold in granules or pellets in slow-release formulations, that can be added directly to the water in the plant. Please follow the manufacturer’s label application instructions.
- Consider planting ornamental bromeliads that might be less favorable habitats for unwanted immature mosquitoes. While appropriate experiments have yet to be done that compare the suitability of popular bromeliad species as habitats for common pest and vector mosquitoes, some general advice is derivable from our current knowledge. Most importantly, a large central tank that can hold abundant water for immature mosquito growth and development is the feature most likely to favor generalist container mosquitoes, such as yellow fever, Asian tiger, or even southern house. Therefore, gardeners worried about favoring pest or vector mosquito species in their yard might consider planting bromeliads with no or reduced central tanks instead, such as Cryptanthus spp. or some species of Tillandsia.
Additionally, to prevent mosquito bites, use mosquito repellent when working outside, cover your skin with long pants, long-sleeved shirts, and close-toed shoes, and cover your doors and windows with screens to keep mosquitoes out of your home. A list of CDC-recommended active ingredients to look for when purchasing an effective mosquito repellent product can be found at
https://www.cdc.gov/zika/prevention/prevent-mosquito-bites.html.
References
Benzing, D. H. 1990. Vascular epiphytes. General biology and related biota. Cambridge, UK: Cambridge University Press.
Benzing, D. H. 2000. Bromeliaceae: Profile of an adaptive radiation. Cambridge, UK: Cambridge University Press. p. 690.
Centers for Disease Control and Prevention (CDC). 2020. “Larvicides.” August 24, 2020. Accessed February 25, 2021. https://www.cdc.gov/mosquitoes/mosquito-control/community/larvicides.html.
Frank, J. H. 2021. “Bromeliad Biota.” Accessed April 21, 2021. https://entnemdept.ufl.edu/frank/bromeliadbiota/index.htm
Frank, J. H. and L. P. Lounibos. 2009. “Insects and Allies Associated with Bromeliads: A Review.” Terrestrial Arthropod Review 1:125–153. doi: 10.1163/187498308X414742.
Frank, J. H., J. P. Stewart, and D. A. Watson. 1988. “Mosquito Larvae in the Axils of the Imported Bromeliad Billbergia pyramidalis in Southern Florida.” Florida Entomologist 71:33–44. https://journals.flvc.org/flaent/article/view/58346.
Gomez, A. 2016. “Miami’s Beloved Shrub Is a Zika Breeding Ground.” U.S.A. Today, September 12, 2016. Accessed February 18, 2021. https://www.usatoday.com/story/news/2016/09/12/zika-miami-bromeliad-mosquito-breeding-ground/90269966/.
Holst, B. K., and H. E. Luther. 2004. “Bromeliaceae (Bromeliad family).” In Flowering Plants of the Neotropics. Edited by N. Smith, S. A. Mori, A. Henderson, D. W. Stevenson, and S. V. Heald. Princeton: Princeton University Press; Princeton, New Jersey, U.S.A: pp. 418–421.
Kitching, R. L. 2000. “Food Webs and Container Habitats: The Ecology and Natural History of Phytotelmata.” Cambridge University Press, Cambridge, England.
Lounibos, L. P., G. F. O’Meara, and N. Nishimura. 2003. “Interactions with native mosquito larvae regulate the production of Aedes albopictus from bromeliads in Florida.” Ecological. Entomology 28:551–558. doi: 10.1046/j.1365-2311.2003.00543.x.
Luther, H. E., and D. H. Benzing. 2009. Native Bromeliads of Florida. Pineapple Press Inc. Sarasota, Florida.
Mocellin, M. G., T. C. Simões, T. F. Silva do Nascimento, M. L. F. Teixera, L. P. Lounibos, and R. Lourenço de Oliveira. 2009. “Bromeliad-inhabiting mosquitoes in an urban botanical garden of dengue endemic Rio de Janeiro – are bromeliads productive habitats for the invasive vectors Aedes aegypti and Aedes albopictus?” Memórias do Instituto Oswaldo Cruz 104:1171–1176. doi: 10.1590/S0074-02762009000800015.
North, G. B., F. H. Lynch, F. D. Maharaj, C. A. Phillips, and W. T. Woodside. 2013. “Leaf Hydraulic Conductance for a Tank Bromeliad: Axial and Radial Pathways for Moving and Conserving Water.” Frontiers in Plant Science. 4:78. doi: 10.3389/fpls.2013.00078.
O’Meara, G. F., M. M. Cutwa, and L. F. Evans. 2003. “Bromeliad-Inhabiting Mosquitoes in South Florida: Native and Exotic Plants Differ in Species Composition.” Journal of Vector Ecology 28:37–46.
Raban, R. R. 2006. “Studies of Aedes albopictus Oviposition and Larval Density, Development and Interactions with Wyeomyia spp. in Southern Florida.” MSc thesis, Department of Entomology and Nematology, University of Florida, Gainesville.
Santos, C. B., G. R. Leite, and A. Falqueto. 2011. “Does (sic) Native Bromeliads Represent Important Breeding Sites for Aedes aegypti in Urbanized Areas?” Neotropical Entomology 40:278–281. doi: 10.1590/s1519-566x2011000200019.
Wilke, A. B. B., C. Vasquez, P. J. Mauriello, and J. C. Beier. 2018. “Ornamental Bromeliads of Miami-Dade County, Florida Are Important Breeding Sites for Aedes aegypti (Diptera: Culicidae)” Parasites and Vectors 11:283–289. doi:10.1186/s13071-018-2866-9.
Xue, R-D, C. Lippi, and L. Drake. 2018. “Species Composition of Mosquitoes and Invertebrates in Common Bromeliad Plant Axils (Family: Bromeliaceae) and the Plant Impacts on Survival of Vector Mosquito Aedes albopictus (Diptera: Culicidae).” China Tropical Medicine 18:6–10. doi: 10.13604/j.cnki.46-1064/r.2018.01.01.