The Featured Creatures collection provides in-depth profiles of insects, nematodes, arachnids and other organisms relevant to Florida. These profiles are intended for the use of interested laypersons with some knowledge of biology as well as academic audiences.
Distribution
Pickleworm is a tropical insect that routinely survives the winter only in south Florida and perhaps south Texas. Pena et al. (1987a) documented the overwintering biology in south Florida, but overwintering has been observed as far north as Sanford, in central Florida, during mild winters. Pickleworm is highly dispersive and invades much of the southeast each summer. North Carolina and South Carolina regularly experience crop damage by pickleworm, but often this does not occur until August or September. In contrast, northern Florida is flooded with moths each year in early June as warm, humid tropical summer weather conditions become firmly established. Although it regularly takes one or two months for the dispersing pickleworms to move north from Florida to the Carolinas, in some years they reach locations as far north as Michigan and Connecticut.
Life Cycle and Description
The pickleworm can complete its life cycle in about 30 days. Over much of its range, multiple and overlapping generations may occur. The number of generations was estimated to be four in Georgia (Dupree et al. 1955) and two or three in North Carolina (Fulton 1947).
Egg
The eggs are minute, measuring only about 0.4–0.6 mm in width and 0.8 mm in length. The shape varies from spherical to flattened. Their color is white initially, but changes to yellow after about 24 hours. The eggs are distributed in small clusters, usually two to seven per cluster. They are deposited principally on the buds, flowers, and other actively growing portions of the plant. Hatching occurs in about four days (Smith 1911). Elsey (1980) estimated egg production to be 300–400 eggs per female.
Larva
There are five instars. Total larval development time averages 14 days. Mean duration (range) of each instar is about 2.5 (2–3), 2 (1–3), 2 (1–3), 2.5 (2–3), and 5 (4–7) days, respectively. Head capsule widths for the five instars are about 0.25, 0.42, 0.75, 1.12, and 1.65 mm, respectively (Smith et al. 1994). Body lengths average 1.6, 2.5, 4.0, 10, and 15 mm during instars 1 through 5, respectively. Young larvae (Figure 1) are nearly white in color with numerous dark gray or black spots. The dark spots are lost at the molt to the fifth instar. Larval color during the last instar is somewhat variable, depending largely on the insect's food source. Prior to pupation, larvae tend to turn a dark copper color. When mature (Figure 2), larvae often attain a length of 2.5 cm (~1 in).

Credit: John L. Capinera, UF/IFAS

Credit: John L. Capinera, UF/IFAS

Credit: John L. Capinera, UF/IFAS
Pupa
Pupation usually occurs in a leaf fold; often, the larva uses dead, dry material to construct its shelter. There is only weak evidence of a cocoon, usually just a few strands of silk. The pupa is elongated, measuring about 13 mm in length and 4 mm in width. It is light brown to dark brown in color, and tapers to a point at both ends. Pupation usually lasts about eight to nine days.
Adult
Emerging moths fly during much of the evening hours, but most flight occurs three to five hours after sundown, with peak flight at approximately midnight (Valles et al. 1991). The female moth produces a pheromone that attracts males, with peak production occurring at five to seven hours after sunset (Valles et al. 1992). Moths are fairly distinctive in appearance (Figure 4). The central portion of both the front and hind wings is a semi-transparent yellow color, with an iridescent purplish reflection. The wings are bordered in dark brown. The wing expanse is about 3 cm. Both sexes often display brushy hairpencils at the tip of the abdomen. Moths are not found in the field during the daylight hours, and probably disperse to adjacent wooded or weedy areas during the heat of the day. Moths do not produce eggs until they are several days old.

Credit: John L. Capinera, UF/IFAS
Host Plants
Pickleworm feeds on both wild and cultivated cucurbit species. Creeping cucumber, Melothria pendula, is considered to be an important wild host. Wild balsam apple, Mormordica chorantia, which has also been reported to be a host, is of questionable significance (Elsey et al. 1985). Summer squash and the winter squash species are good hosts. Pumpkin is of variable quality as a host, probably because pumpkins have been bred from several Cucurbita species. The Cucumis species, cucumber, gerkin, and cantaloupe, are attacked but not preferred. Among all cucurbits, summer squash is most preferred, and most heavily damaged. Cultivars vary widely in susceptibility to attack, but truly resistant cultivars are unknown (Dilbeck et al. 1974).
Damage
Pickleworm may damage summer and winter squash, cucumber, cantaloupe, and pumpkin. Watermelon is an unusual host. The blossom is a favored feeding site, especially for young larvae. In plants with large blossoms, such as summer squash, larvae may complete their development without entering fruit. They may also move from blossom to blossom, feeding and destroying the plant's capacity to produce fruit. Very often, however, the larva burrows into the fruit. The larva's entrance is marked by a small hole, through which frass is extruded. The presence of the insect makes fruit unmarketable, and fungal or bacterial diseases often develop once entry has occurred. When all blossoms and fruit have been destroyed, larvae will attack the vines, especially the apical meristem. Cantaloupe is not a preferred host, and larvae often seem reluctant to burrow into the fruit. Rather, they feed on the surface or "rind", causing scars. Thus, pickleworm is sometimes referred to as "rindworm," though their feeding is not restricted to the surface, and they sometimes burrow into the fruit. When offered a choice among cantaloupe, squash, and watermelon for egg laying under greenhouse conditions, female pickleworm moths preferred squash over watermelon and cantaloupe (Leiner and Spafford 2016a). Newly hatched larvae were less particular, choosing equally among squash, cantaloupe, and watermelon (Leiner and Spafford 2016b).
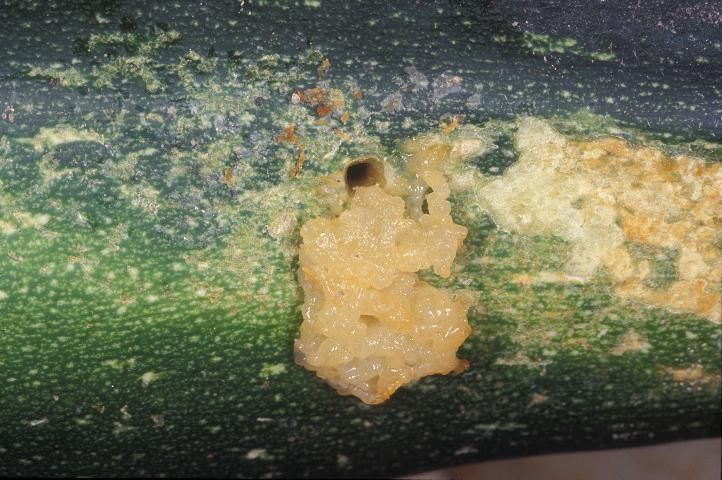
Credit: Lyle Buss, UF/IFAS
Natural Enemies
Pickleworm has several natural enemies, but none reliably suppress damage. Generalist predators such as Calosoma spp. and Harpalus (both Coleoptera: Carabidae), the soldier beetle Chauliognathus pennsylvanicus DeGeer (Coleoptera: Cantharidae), and the red imported fire ant Solenopsis invicta Buren (Hymenoptera: Formicidae) have been reported to be important mortality factors. Also, several parasitoids are known, including Apanteles sp., Hypomicrogaster diaphaniae (Muesebeck), Pristomerus spinator (Fabricius) (all Hymenoptera: Braconidae), Casinaria infesta (Cresson), Temelucha sp. (both Ichneumonidae), and undetermined trichogrammatids (Pena et al. 1987b, Capinera 1994). The braconid Cardiochiles diaphaniae Marsh (Hymenoptera: Braconidae) has been imported from Colombia and released into Florida and Puerto Rico in an attempt to obtain higher levels of parasitism (Smith et al. 1994), and apparently has established in the latter location.
Management
Sampling
It is very difficult to scout for this insect and predict its appearance. Moths are not attracted to light traps, and although pheromone traps have had some success experimentally (Elsey et al. 1991, Valles et al. 1991), pheromone lures are not currently available commercially. Brewer and Story (1987) developed sampling plans for pickleworm larvae in squash. They suggested that the most reliable sampling unit is the large green staminate flower bud. However, the small eggs, night-flying behavior, and inability to trap the insect reliably lead most growers to depend on preventative applications of insecticides.
Insecticides
Cucurbit producers in areas where pickleworm damage is likely to occur usually apply chemical insecticides from the onset of fruiting through harvest. The internal feeding behavior of larvae, which is so difficult to detect at harvest, causes particular emphasis on prevention of damage. In areas that are on the fringe of the normal range, there are many seasons when damage will not occur, but producers apply insecticides as a preventative measure because prediction of occurrence is so difficult.
Pollinators, particularly bees, are very important in cucurbit production, and insecticide application can interfere with pollination by killing bees. If insecticides are to be applied when blossoms are present, it is advisable to use insecticides with little residual activity, and to apply insecticides late in the day, when honeybee activity is minimal.
See the Vegetable Production Handbook
Biological Control
The entomopathogenic nematode Steinernema carpocapsae has been shown to effectively suppress pickleworm injury in squash (Shannag et al. 1994). Nematode survival is quite good in large-blossomed squash, where the nematodes can kill the young pickleworm before it burrows into the fruit. This approach is probably ineffective for species with small, open blossoms such as cucumber, however, because the nematodes die quickly when exposed to sunlight. Bacillus thuringiensis will kill pickleworm but is usually not recommended because the internal feeding behavior puts the feeding larvae beyond the reach of a stomach-active toxin.
Cultural Practices
It is possible to cover plants with screen or row covers to prevent moths from depositing eggs on the foliage (Webb and Linda 1992). However, because the plants must be pollinated, usually by honey bees, some allowance must be made to leave the plants uncovered. Given the night-flying behavior of the moths and the daytime activities of honeybees, this is not a difficult task on a small planting but is prohibitive on large acreage.
Some growers are able to prevent plant injury through careful timing of their cropping cycle. By planting early, it is often possible to harvest part of the crop before pickleworms appear.
Smith (1911) reported that squash could be used as a trap crop to keep pickleworm from attacking cantaloupe, a less preferred host. He recommended that destruction of squash blossoms, or even the entire plant, be done periodically to keep pickleworms from exhausting the food supply and then moving onto adjacent cantaloupes. In contrast, Dupree et al. (1955) reported unsatisfactory results with trap cropping.
Selected References
Brewer, M.J. and R.N. Story. 1987. Larval spatial patterns and sequential sampling plan for pickleworm, Diaphania nitidalis (Stoll) (Lepidoptera: Pyralidae), on summer squash. Environmental Entomologist 16:539–544.
Capinera, J.L. 1994. Pickleworm and melonworm. Pages 140–145 in D. Rosen, F.D. Bennett, and J.L. Capinera (eds.). Pest Management in the Subtropics: Biological Control -- A Florida Perspective. Intercept, Andover, United Kingdom.
Capinera, J.L. 2001. Handbook of Vegetable Pests. Academic Press, San Diego. 729 pp.
Dilbeck, J.D., J.W. Todd, and T. Canerday. 1974. Pickleworm resistance in Cucurbita. Florida Entomologist 57:27–32.
Dupree, M., T.L. Bissell, and C.M. Beckham. 1955. The pickleworm and its control. Georgia Agricultural Experiment Station Bulletin N.S. 5:1–34.
Elsey, K.D. 1980. Pickleworm: mortality on cucumbers in the field. Environmental Entomologist 9:806–809.
Elsey, K.D., J.A. Klun, and M. Schwarz. 1991. Forecasting pickleworm (Lepidoptera: Pyralidae) larval infestations using sex pheromone traps. Journal of Economic Entomology 84:1837–1841.
Elsey, K.D., J.E. Pena, and V.H. Waddill. 1985. Suitability of potential wild hosts of Diaphania species in southern Florida. Florida Entomologist 68:682–686.
Gonring, A.H. R., M.C. Picanço, J. C. Zanuncio, M. Puiatti, and A.A. Semeão. 2002. Natural biological control and key mortality factors of the pickleworm, Diaphania nitidalis Stoll (Lepidoptera: Pyralidae), in cucumber. Biol. Agric. Hort. 20:365–380.
Kelsheimer, E.G. 1949. Control of insect pests of cucumber and squash. Florida Agricultural Experiment Station Bulletin 465. 15 pp.
Leiner, R.C. and H. Spafford 2016a. Oviposition preferences of pickleworm (Lepidoptera: Crambidae) in relation to a potential push-pull cropping management approach. Environmental Entomology 45: 677–684.
Leiner, R.C. and H. Spafford 2016b. Pickleworm (Diaphania nitidalis Cramer) neonate feeding preferences and the implications for a push-pull management system. Insects 7: 32; DOI:10.3390/insects7030032
Peña, J.E., V.H. Waddill, and K.D. Elsey. 1987a. Population dynamics of the pickleworm and the melonworm (Lepidoptera: Pyralidae) in Florida. Environmental Entomologist 16:1057–1061.
Peña, J.E., V.H. Waddill, and K.D. Elsey. 1987b. Survey of native parasites of the pickleworm, Diaphania nitidalis Stoll, and melonworm, Diaphania hyalinata (L.) (Lepidoptera: Pyralidae), in southern and central Florida. Environmental Entomologist 16:1062–1066.
Smith, H.A., J.L. Capinera, J.E. Peña,, and B. Linbo-Terhaar. 1994. Parasitism of pickleworm and melonworm (Lepidoptera: Pyralidae) by Cardiochiles diaphaniae (Hymenoptera: Braconidae). Environmental Entomologist 23:1283–1293.
Smith, R.I. 1910. Insect enemies of cantaloupes, cucumbers and related plants. North Carolina Agricultural Experiment Station Bulletin 205. 40 pp.
Smith, R.I. 1911. Two important cantaloupe pests. North Carolina Agricultural Experiment Station Bulletin 214:101–146.
Valles, S.M., J.L. Capinera, and P.E.A. Teal. 1991. Evaluation of pheromone trap design, height, and efficiency for capture of male Diaphania nitidalis. (Lepidoptera: Pyralidae) in a field cage. Environmental Entomologist 20:1274–1278.
Valles, S.M., R.R. Heath, and J.L. Capinera. 1992. Production and release of sex pheromone by Diaphania nitidalis (Lepidoptera: Pyralidae): Periodicity, age, and density effects. Annals of the Entomological Society of America 85:731–735.
Webb, S.E. and S.B. Linda. 1992. Evaluation of spunbonded polyethylene row covers as a method of excluding insects and viruses affecting fall-grown squash in Florida. Journal of Economic Entomology 85:2344–2352.