Introduction
Utetes anastrephae (Viereck) is the only species of Utetes recorded repeatedly from several species of Anastrepha, a genus of tephritid fruit flies that contains several major pest species (Ovruski et al. 2000). It is also the only Ananstrepha-attacking Utetes species (see Host Table below) native to the continental United States (Wharton 1997).
Synonymy
Utetes anastrephae was originally described as Opius anastrephae by Viereck in 1913. In 1977, Fischer transferred it to the genus Bracanastrepha, and it was finally placed in the genus Utetes by Wharton (1988). Bracanastrepha argentina Brèthes, 1924 and Opius mombinpraeoptantis (Fischer 1966) are synonyms (Wharton and Marsh 1978).
Distribution
Utetes anastrephae has a wide distribution that extends from Florida to Argentina (Wharton and Marsh 1978, Sivinski et al. 1997). In addition to parasitizing several Ananstrepha spp., both pests and non-pests (Wharton and Gilstrap 1983), it has also been collected from the Mediterranean fruit fly, Ceratitis capitata, in Argentina (Nasca 1973).
Description
Adult: "Utetes anastrephae" may actually be a complex of closely related species, with subtle differences in ovipositor length, body sculpture, and host preferences (Wharton 1997). A number of color morphs have been obtained from the same host-collections. This species has a relatively short ovipositor ~ 1.6 mm in length (Sivinski et al. 2001). The absence of an occipital carina (an upside-down U-shaped ridge on the back or posterior surface of the head) is characteristic of the Utetes anastrephae species group, as is a sharp ridge located basal-medially on the hind tibia.

Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida

Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida
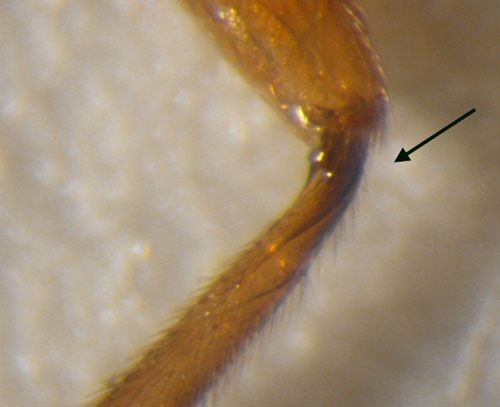
Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida
The Cu2 submarginal cell of the forewing is five-sided and relatively long compared to Cu1 and Cu3 (Wharton and Marsh 1978).
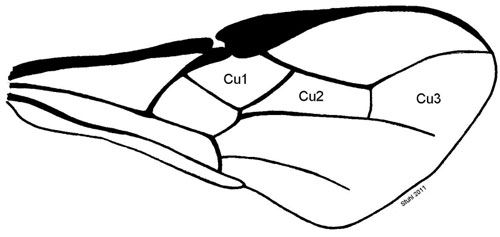

Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida
Life Cycle and Behavior
Utetes anastrephae is a larval-prepupal synovigenic (produce eggs over the life of the adult), endoparasitic koinobiont (parasitoid allows the host to continue development and does not kill the host until the parasitoid larva pupates) that develops well in late instar larvae. It forages for larvae in ripe fruit hanging on tree branches (Ovruski et al. 2000) and the female inserts a single egg inside the body of the fly larvae. Upon hatching, the parasitoid larva remains in the first instar stage until the host begins to pupate. Development time from egg to adult parasitoid is temperature dependent, but usually takes about two weeks. Females are attracted to fruit volatiles in their search for food and fly hosts. Host location within the fruit is mediated by antennation (sensing information by touching antennae) and perhaps probing with the ovipositor to detect compounds unique to larval hosts (Stuhl et al. 2011). It is assumed that fruit juices and other plant-produced substances such as extrafloral nectars are the food source of this parasitoid.
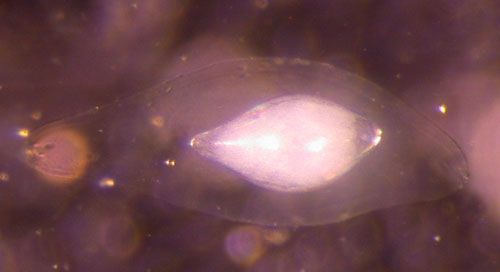
Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida

Credit: Charles Stuhl, USDA-ARS-CMAVE Gainesville, Florida
Economic Importance
Utetes anastrephae contributes to the mortality of the Caribbean fruit fly, Anastrepha suspensa (Loew), in Florida and in Mesoamerica, inflicting very high levels of parasitism on the West Indian fruit fly, Anastrepha obliqua (Macquart). It occupies niches, e.g., the exploitation of hosts in native fruit trees in Mexico, that commonly imported fruit fly parasitoids, such as braconid Diachasmimorpha longicaudata (Ashmead), may be less suited to exploit (Lopez et al., 1999).
Selected References
Aluja M, Piñero J, López J, Ruiz C, Zuniga A, Piedra E, Díaz-Fleischer F, Sivinski J. 2000. New host plant and distribution records in Mexico for Anastrepha spp., Toxotrypana curvicauda Gerstacker, Rhagoletis zoqui Bush, Rhagoletis sp., and Hexachaeta spp (Diptera : Tephritidae). Proceedings of the Entomological Society of Washington 102: 802–815.
Aluja M, Rull J, Sivinski J, Norrbom L, Wharton RA, Macías-Ordoñez R, Díaz-Fleischer F, López M. 2003. Fruit Flies of the Genus Anastrepha (Diptera: Tephritidae) and associated native parasitoids (Hymenoptera) in the tropical rainforest biosphere reserve of Montes Azules, Chiapas, Mexico. Environmental Entomology 32: 1377–1385.
Brethes J. 1924. Varios Himenopteros de la America de Sud. Nunquam Otiosus 1924: 6–16, 145–175.
Fischer M. 1977. Hymenoptera, Braconidae (Opiinae II-Amerika) Das Tierreich 96: 1–1001.
Fischer M. 1966. Neue neotropische Opius-arten aus dem U.S. National Museum, Washington. (Hymenoptera, Braconidae). Beiträge zur Entomologie 16(1/2): 84–150.
López M, Aluja M, Sivinski J. 1999. Hymenopterous larval pupal and pupal parasitoids of Anastrepha flies (Diptera: Tephritidae) in Mexico. Biological Control 15: 119–129.
Nasca AJ. 1973. Parasitos de 'moscas de los frutos' establecidos en algunas zonas de Tucuman. Revista Agronomica del Noroeste Argentino 10: 31–43.
Ovruski S, Aluja M, Sivinski J, Wharton R. 2000. Hymenopteran parasitoids of fruit-infesting Tephritidae (Diptera) in Latin America and the southern United States: Diversity, distribution, taxonomic status and their use in fruit fly biological control. Integrated Pest Management Review 5: 81–107.
Sivinski J, Aluja M, López M. 1997. Spatial and temporal distributions of parasitoids of Mexican Anastrepha species (Diptera: Tephritidae) within the canopies of fruit trees. Annals of the Entomological Society of America 90: 604–618.
Sivinski J, Vulinec K, Aluja M. 2001. Ovipositor length in a guild of parasitoids (Hymenoptera: Braconidae) attacking Anastrepha spp. fruit flies (Diptera: Tephritidae) in southern Mexico. Annals of the Entomological Society of America 94: 886–895.
Stuhl C, Sivinski J, Teal P, Paranhos B, Aluja M. 2011. A compound produced by fruigivorous Tephritidae (Diptera) larvae promotes oviposition behavior by the biological control agent Diachasmimorpha longicaudata (Hymenoptera: Braconidae). Environmental Entomology 40: 727–736.
Viereck HL. 1913. Descriptions of ten new genera and twenty-three new species of Ichneumon-flies. Proceedings of the United States National Museum 44: 555–568.
Wharton RA. 1997. Generic relationships of opiine Braconidae (Hymenoptera) parasitic on fruit-infesting Tephritidae (Diptera). Contributions of the American Entomological Institute 30: 1–53
Wharton RA. 1988. Classification of the braconid subfamily Opiinae (Hymenoptera). Canadian Entomologist 120: 333–360.
Wharton RA, Gilstrap FE. 1983. Key to and status of opiine braconid (Hymenoptera) parasitoids used in biological control of Ceratitis and Dacus s. l. (Diptera: Tephritidae) Annals of the Entomological Society of America 76: 721–742
Wharton RA, Marsh PM. 1978. New World Opiinae (Hymenoptera: Braconidae) parasitic on Tephritidae (Diptera). Journal of the Washington Academy of Science 68: 147–167.