Equine coronavirus (ECoV) is a contagious virus causing fever and illness in horses throughout the continental United States. The first outbreaks of ECoV were reported from 2009 to 2011 in Japan, Europe, and the US. ECoV primarily affects the intestinal system in adult horses and causes lethargy, fever, anorexia (lack of appetite), and occasionally colic and diarrhea. The role of ECoV as a cause of disease in foals is still unclear because the virus can be detected in both healthy and sick foals. This article highlights current knowledge regarding the transmission, clinical signs, and management of ECoV in horses.
Prevalence
Increased awareness and availability of diagnostic testing have led to steadily increasing numbers of confirmed ECoV cases reported by veterinary diagnostic laboratories since 2010. In a recent study, 9.6% of adult healthy horses from the United States were found to have blood antibodies to ECoV, suggesting that many horses are exposed to the virus (Kooijman et al. 2017). While exposure to ECoV appears to be relatively common, only some horses will develop signs of illness, and only some will shed infective virus in their manure. In a recent study of horses hospitalized for gastrointestinal disease, ECoV was found in less than 1% (1/258) of manure samples (Sanz et al. 2019).
Transmission
Transmission of ECoV is fecal-oral and occurs when horses come into contact with manure from infected horses. ECoV cases occur year-round, although many cases are diagnosed during the winter months. Increased horse-to-horse contact due to indoor housing of horses during the winter may contribute to the seasonal variation in cases. Additionally, the virus may survive better in the environment when ambient temperatures are low. Contaminated stalls, muck forks, manure spreaders, thermometers, hands, and clothing can also contribute to the spread of ECoV. A small percentage of infected horses may also shed virus in their nasal secretions; however, this is not thought to be a primary route of transmission between horses (Pusterla et al. 2015; Pusterla et al. 2019). During farm outbreaks, horses that appear normal may shed virus and serve as “silent carriers” of ECoV within the herd.
Clinical Disease
ECoV causes fever, lethargy, and intestinal disease in adult horses (Figure 1). Most horses develop clinical signs within 2–3 days after infection (Schaefer et al. 2018). Clinical signs are largely from intestinal inflammation, and may also include red/toxic mucous membranes and elevated heart rate and respiratory rates (Figure 2). Bloodwork from infected horses often reveals low white blood cell counts (leukopenia) and signs of dehydration. The severity of illness is generally mild, although more severe clinical signs and occasional deaths have been reported. Cases may occur in individual horses or as herd outbreaks. The percentage of horses that become infected within a herd outbreak has been reported to be between 10% and 83% (Pusterla et al. 2013).
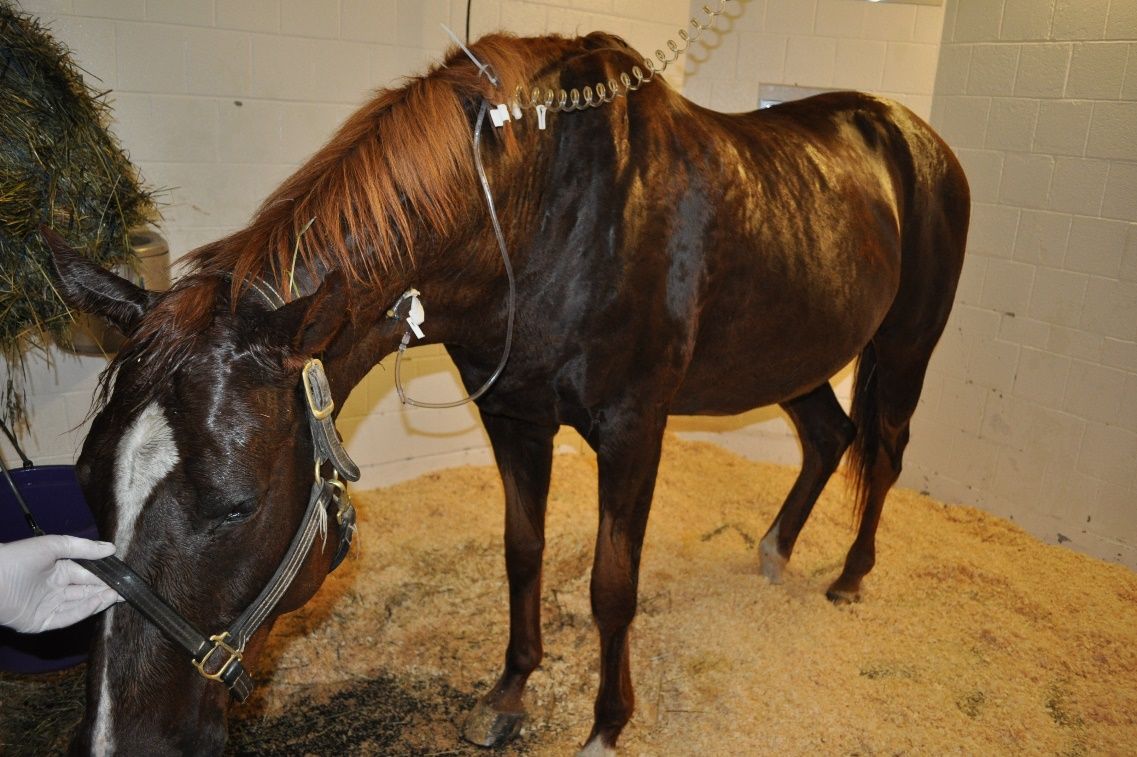
Credit: Sally DeNotta, UF
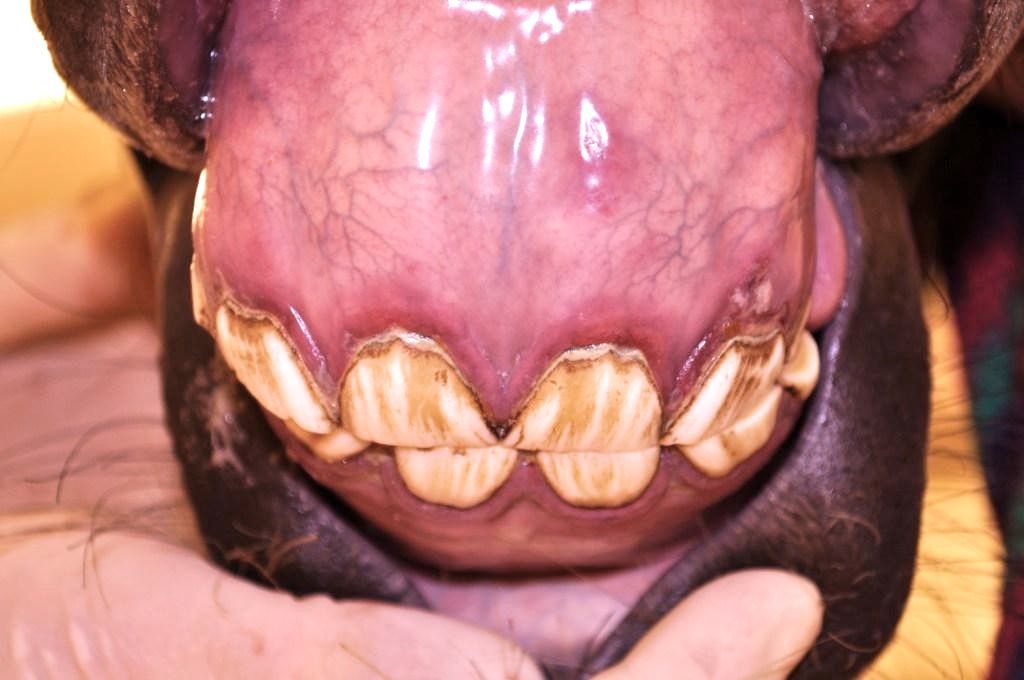
Credit: Sally DeNotta, UF/IFAS
Clinical signs of ECoV as reported from 20 outbreaks (Pusterla et al. 2013; Pusterla et al. 2018) include:
- Anorexia (no appetite) (97%)
- Lethargy (88%)
- Fever (83%)
- Low white blood cell count (leukopenia) (73%)
- Soft manure and/or diarrhea—inconsistent finding (23%)
- Colic (19%)
Occasionally, neurologic signs may be observed in horses with ECoV secondary to elevated concentrations of ammonia in the blood. These signs may include aimless wandering, depressed attitude, incoordination, head pressing, or seizures. For unknown reasons, miniature horses appear to be at higher risk of neurologic complications from ECoV infection (Figure 3).
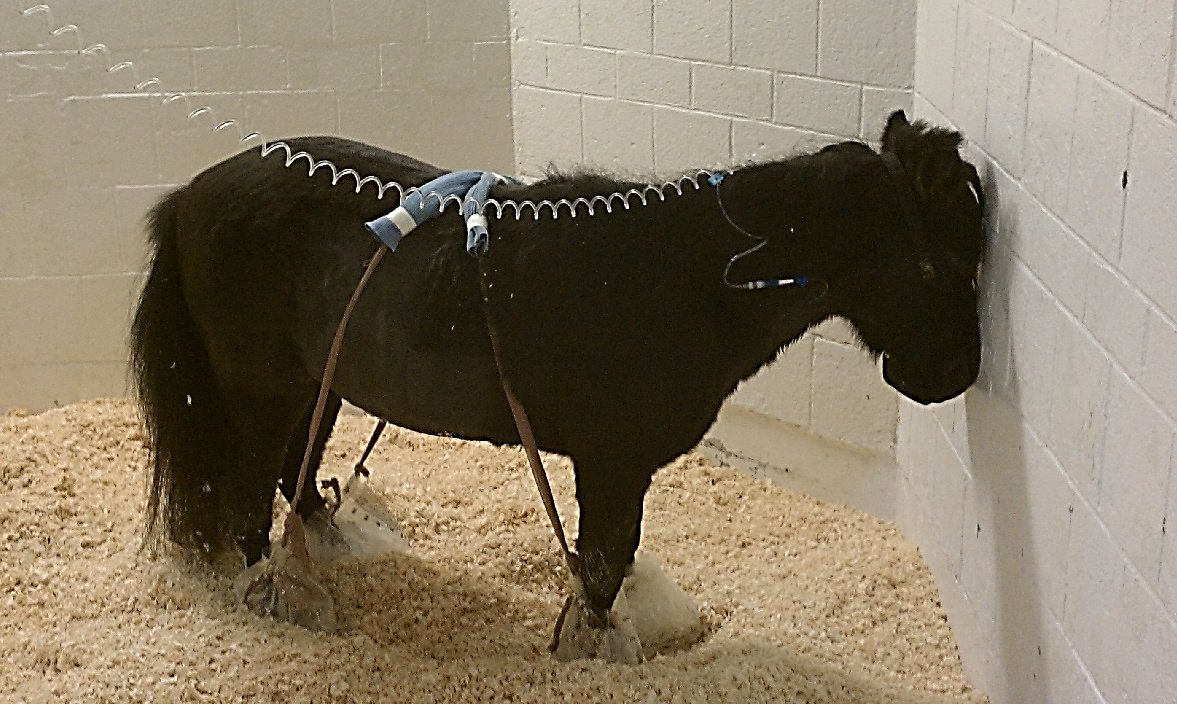
Credit: Sally DeNotta, UF/IFAS
Diagnostic Sampling, Testing, and Handling
The diagnosis of ECoV infection is confirmed by detecting ECoV in feces via PCR testing. Several veterinary laboratories offer testing for ECoV on fresh or frozen feces samples. In experimental infection studies, horses began shedding ECoV in their feces 3–4 days following infection, and peak viral shedding occurred 3–4 days after the development of clinical signs (Schaefer et al. 2018). Postmortem diagnosis of ECoV can be achieved by PCR on feces, small intestinal contents, or tissue from the small intestine collected at postmortem examination.
Under natural conditions, fecal shedding of ECoV from infected horses has been reported to range from 3 to 25 days. However, the authors have followed horses that continue to shed the virus for more than 30 days following outbreaks, including horses that remained asymptomatic the entire time they were shedding virus. In a small experimental infection study, horses shed from days 3 to 15 following infection (Schaefer et al. 2018). In a recent outbreak in Florida, horses in stalls adjacent to a clinical ECoV case never developed fever or clinical signs, but had low white blood cell counts and were PCR positive for ECoV on fecal testing. At least one of the horses involved in this outbreak never showed clinical signs and remained PCR positive on feces for 6 weeks or longer (DeNotta, unpublished data).
Treatment
There is no specific treatment for ECoV. Supportive therapies with anti-inflammatories and occasionally fluid therapy can be helpful for reducing fever and correcting dehydration. In horses with fevers and intestinal inflammation, digital cryotherapy (ice boots) may be helpful for preventing laminitis. More severe cases and those with neurologic symptoms may require hospitalization and intensive medical care. Fecal transfaunation (administration of manure from a healthy horse to a horse with intestinal disease) has not been specifically evaluated for the treatment of ECoV, but it may be beneficial for ECoV-infected horses with diarrhea.
Prognosis
The prognosis for ECoV is generally good, with most infections being mild and self-limiting. Exposure to the virus can result in up to 85% infection rate, but most animals do not show clinical signs. Mortality is generally low, with 96% of horses surviving to discharge in a recent report of hospitalized cases of ECoV (Berryhill et al. 2019).
Prevention and Biosecurity
Prevention of ECoV infection involves minimizing contact between horses of unknown infection status and maintaining high standards of sanitation in all equine facilities, especially when traveling. Manure should be disposed of in a location where it cannot contaminate pastures, paddocks, or drinking water. New horses in equine facilities should be quarantined for 3 weeks before integration into the resident population. There are currently no vaccines for ECoV. Vaccines have been successfully used to prevent bovine coronavirus (BoCV), a closely related beta coronavirus, in herds; however, these vaccines have not yet been evaluated in horses.
Any horse with fever should be considered potentially infected with ECoV and isolated from other horses immediately. Horses found to be positive for ECoV via fecal PCR should remain in isolation, and strict biosecurity measures and manure management should be implemented to prevent the spread of infection to other horses in the vicinity. Other horses on the property should be monitored for fevers (assess and record temperature at least twice each day) and/or leukopenia. Farm personnel working with horses that are shedding ECoV should wear disposable gloves and personal protective equipment (gowns or dedicated coveralls, boot covers) and handle infected horses last and as infrequently as possible. Diligent hand washing is also important, and a 70% ethanol hand sanitizer should be used when moving between horses. Horses should remain in strict quarantine until confirmed to be fecal PCR negative. Typically, shedding periods range from 2 to 21 days, but may be longer in some cases. Therefore, isolating infected horses for a predetermined period and then presuming the horse has stopped shedding is not recommended.
Potential for Transmission to Other Species
Although coronaviruses affect many species worldwide, ECoV has only been reported in horses. Transmission of ECoV from horses to other species has not been documented.
Additional Information
Detailed information on ECoV including up-to-date reports of ECoV cases by county and state and information regarding infectious disease control are available through the Equine Disease Communication Center at http://www.equinediseasecc.org/.
References
Berryhill, E., K. Magdesian, M. Aleman, and N. Pusterla. 2019. “Clinical Presentation, Diagnostic Findings, and Outcome of Adult Horses with Equine Coronavirus Infection at a Veterinary Teaching Hospital: 33 Cases (2012–2018).” Vet. J. 248:95–100.
Fielding, C., J. Higgins, J. Higgins, S. McIntosh, E. Scott, F. Giannitti, A. Mete, and N. Pusterla. 2015. “Disease Associated with Equine Coronavirus Infection and High Case Fatality Rate.” J. Vet. Intern. Med. 29:307–310.
Kooijman, L. J., K. James, S. M. Mapes, M. J. Theelan, and N. Pusterla. 2017. “Seroprevalence and Risk Factors for Infection with Equine Coronavirus in Healthy Horses in the USA.” Vet. J. 220:91–94.
Pusterla, N., N. Holzenkaempfer, S. Mapes, and P. Kass. 2015. “Prevalence of Equine Coronavirus in Nasal Secretions from Horses with Fever and Upper Respiratory Tract Infection.” Vet. Rec. 177:289.
Pusterla, N., K. James, S. Mapes, and F. Bain. 2019. “Frequency of Molecular Detection of Equine Coronavirus in Faeces and Nasal Secretions in 277 Horses with Acute Onset of Fever.” Vet. Rec. 184(12): 385.
Pusterla, N., S. Mapes, C. Wademan, A. White, R. Ball, K. Sapp, P. Burns, et al. 2013. “Emerging Outbreaks Associated with Equine Coronavirus in Adult Horses.” Vet. Micro. 162:228–231.
Pusterla, N., R. Vin, C. Leutenegger, L. Mittel, and T. Divers. 2018. “Enteric Coronavirus Infection in Adult Horses.” Vet. J. 231:13–18.
Sanz, M. G., S. Kwon, N. Pusterla, J. R. Gold, F. Bain, and J. Evermann. 2019. “Evaluation of Equine Coronavirus Fecal Shedding among Hospitalized Horses.” J. Vet. Intern. Med. 33(2): 918–922.
Schaefer, E., C. Harms, M. Viner, S. Barnum, and N. Pusterla. 2018. “Investigation of an Experimental Infection Model of Equine Coronavirus in Adult Horses.” J. Vet. Intern. Med. 32(6): 2099–2104.