Abstract
Florida is experiencing many human and climate-related changes to the aquatic environment. These impacts can lead to changes among fish populations as habitat is lost or altered, impacting the survival, growth, and/or recruitment of those populations. Recruitment is important because it influences how many adult fish are later available for spawning (and continuing the population). Factors independent of, or dependent on, fish density (number per area) can impact recruitment. Increased fish densities can intensify ecological interactions, such as predation and competition for food and habitat. These interactions can lead to increased mortality. However, survival during the recruitment period also depends on factors unrelated to the density of fish, such as environmental factors, including water quality and habitat. Understanding these factors and how they are related can be important for considering the potential effects of ongoing climate change and for restoration and management of water quality and habitat. The purpose of this publication is to provide the necessary background information to consider these effects on fish populations. This publication should be useful to the public interested in understanding more about some of the most important factors affecting fish populations. This publication should be especially useful to Extension agents and management agency personnel who would like an overview of these topics prior to engaging with stakeholders.
Introduction
Fish populations are constantly changing. They change due to shifts in fishing harvest and natural mortality. They are also influenced by variations in the environment, including impacts of climate change, human development, natural environmental shifts (such as seasonal weather patterns), as well as by randomness in populations genetics (called demographic stochasticity). Fish populations in Florida may be especially subject to faster rates of environmental change. This is because a growing human population increasingly affects coastal areas and coincides with a deepening climate crisis that is potentially altering typical rainfall patterns and exacerbating storms, influencing Florida’s estuarine and coastal areas. These changes may well affect one of the most critical fish population life history stages, namely the recruitment stage. This publication describes some of the factors that can affect the recruitment of finfish populations. Specifically, it talks about processes that are dependent on the fish population density—the abundance of fish within an area—called “density-dependent processes,” as well as processes that are unrelated to fish population density, called “density-independent processes.” Finfish recruitment can be complicated, and it is challenging to understanding how density-dependent and -independent processes operate at the same time.
Scientists believe recruitment to be one of the most important life history stages for fish. Recruitment generally refers to the early life stage that young fish must survive to become a sub-adult or adult (Figure 1) (Doherty and Williams 1988; Peterson et al. 2003). The recruitment stage differs from other fish life stages because much of the natural mortality fish experience during the recruitment period is density-dependent. Density-dependent mortality means that mortality increases as fish density increases. This is because when there are high densities of fish, competition for resources increases, so the availability of things like food and refuge goes down, decreasing the chances of survival (Camp et al. 2020). Mortality also decreases as fish density decreases. Density-dependent mortality is important because it provides some population stability. When there are fewer fish overall (low density) entering the recruitment period, those juveniles enjoy lower mortality and survive to adulthood better. As long as the overall adult population stays above a critical minimum abundance, this can help populations recover from overfishing or other sources of high mortality. When there are more fish overall (greater density) entering the recruitment period, mortality will increase, and a smaller proportion of fish will survive to reach adulthood. In this way, density-dependent mortality keeps populations from growing out of control. Together, density-dependent processes help bring (some) stability and resilience to populations. But multiple factors can limit the capacity for populations to successfully “compensate” for density differences. There may be fluctuations in the number of eggs (related to the number of spawning adults) or environmental changes, for instance. These factors are often density-independent, affecting all fish regardless of their abundance. Most importantly, because density-dependent survival generally ends after the recruitment period, recruitment effectively “sets” year-classes for the population (Loreznen and Camp 2019). In other words, the number of juvenile fish that survive recruitment (referred to as “recruits”) should be directly related to the size of the adult population (Anderson 1988; Sogard 1997). Recruitment directly influences the number of fish available to catch and spawn. More information on recruitment can be found in FA222 and FA234.

Credit: Florida Sea Grant
Influences on Recruitment
Density-Independent Processes
Like all biological processes, recruitment is influenced by many environmental factors, several of which do not depend on fish density. These are often abiotic (nonliving) factors. Temperature, salinity, and dissolved oxygen concentration can cause mortality before, during, and after fish are in the recruitment stage. Many fish have strict water quality tolerances, so temperature or salinity levels outside the tolerance threshold of a species can limit survival, regardless of density. Because of this, environmental factors can potentially have a greater effect on mortality than density-dependent factors, because young fish can rarely live outside their water quality tolerances. For example, if the minimum temperature tolerance for juvenile common snook is about 48 degrees Fahrenheit, extended water temperatures less than 48 degrees could kill all juvenile snook in the affected area, regardless of density.
Water quality characteristics like dissolved oxygen and salinity themselves change because of another important variable: water flow. Flow is the movement of both fresh and saline water through space, and changes in flow relate to the speed or amount of moving water. Water flow in coastal rivers has a massive effect on salinity in estuaries and coastal waters because changes in river flow influence the amount of freshwater entering the estuary and marine environment. Water flowing from rivers can sometimes be low in oxygen or have excess nutrients, chemicals, and sediment that can lead to low oxygen and/or clarity, especially in certain parts of Florida following heavy rains. Decreased water clarity can affect fish ability to find prey or avoid predators and can also impact habitats important to fish, like submerged aquatic vegetation (e.g., seagrasses). Excess nutrients can lead to harmful algal blooms, which may result in low oxygen and fish kills in extreme situations. When flow does not directly affect fish survival, it can still dictate where fish larvae move while they are too young to swim or too small to swim against a current. Water flow can transport fish to different habitat types that may or may not be suitable for the survival of that species, depending on, for example, the water quality or food type available at that location. So, transportation of young fish by water flow heavily influences successful recruitment. The effect of changing water flow on recruitment is not straightforward. Changing flow velocities or changes to river flow volume can have overall positive, negative, or neutral impacts on fish recruitment, depending on the species, timing, and location (Bonvechio and Allen 2005). For this reason, a lot of past and current research (in Florida and elsewhere) focuses on how freshwater flow affects the eventual recruitment of coastal and estuarine fish.
Water temperature, salinity, dissolved oxygen, and water flow are not the only density-independent variables that affect fish survival during the recruitment period, but they are some of the variables that scientists have commonly shown to have strong effects on fish survival. A change in one of these variables can have a range of effects on fish recruitment. Effects can be positive or negative, weak or strong, depending on the intensity of the environmental change and the fish species. It is important to understand that these variables can affect fish recruitment all by themselves, without any interacting density effects. This means that these variables should be monitored, and changes ought to trigger research efforts to assess the effects on fish.
Density-Dependent Processes
Assuming the environmental conditions are not limiting survival, juveniles must then survive the interactions they have with other individuals: the biotic factors. These competitive species interactions are the drivers of density-dependent mortality in the recruitment phase. In high densities, there is greater competition for resources among fish, primarily for refuge habitat and food (Anderson 1988). It is logical that small fish will need food to not starve and will need refuge to not be eaten by predators. Forage and refuge resources are understood to be intricately related. The more food young fish eat, the faster they grow, and as they grow, their risk of predation and mortality decreases. At the same time, fish are more vulnerable to predation while they are foraging, so young fish need to compete to get to areas that provide plenty of food near good refuge (Ahrens et al. 2012). In lower fish densities, a greater proportion of the small fish will be able to use these good refuge and forage spaces, increasing the survival rate. In higher densities, a lower proportion will be able to use those spaces, and the survival rate will decline. This also means that if the amount of suitable fish habitat changes, the total number of fish surviving recruitment may change as well. This is one of the reasons that habitats that provide refuge are so important for recruiting fish (Figure 2). In the scientific literature, these important habitats are often referred to as “nursery” habitats (Beck et al. 2001).

Credit: Florida Sea Grant
Density-dependent competition (and mortality) depends not only on the quantity of resources, however. The quality of environmental attributes can also interact with and alter the fish-to-fish interactions that lead to density-dependent mortality. Fish interactions with habitat include both biotic and abiotic factors, as well as density-dependent and density-independent processes. These characteristics include not only the physical habitat structures themselves, but also the physical and chemical aspects of the habitat location and the abundance of food resources present. For example, a decrease in the abundance of salt marsh in an estuary could directly increase the competition for that habitat among the fish that need it. But also, a change in the water salinity or dissolved oxygen can force fish into a smaller marsh area than before, even if the same quantity of the marsh itself is still present. These conditions increase density-dependent competition for the portion of the marsh that lies within the area with better water quality. Both of these scenarios require recruiting fish to compete for food and refuge in a smaller area than before, intensifying density-dependent effects and decreasing survival. This example is illustrated in Figure 3.
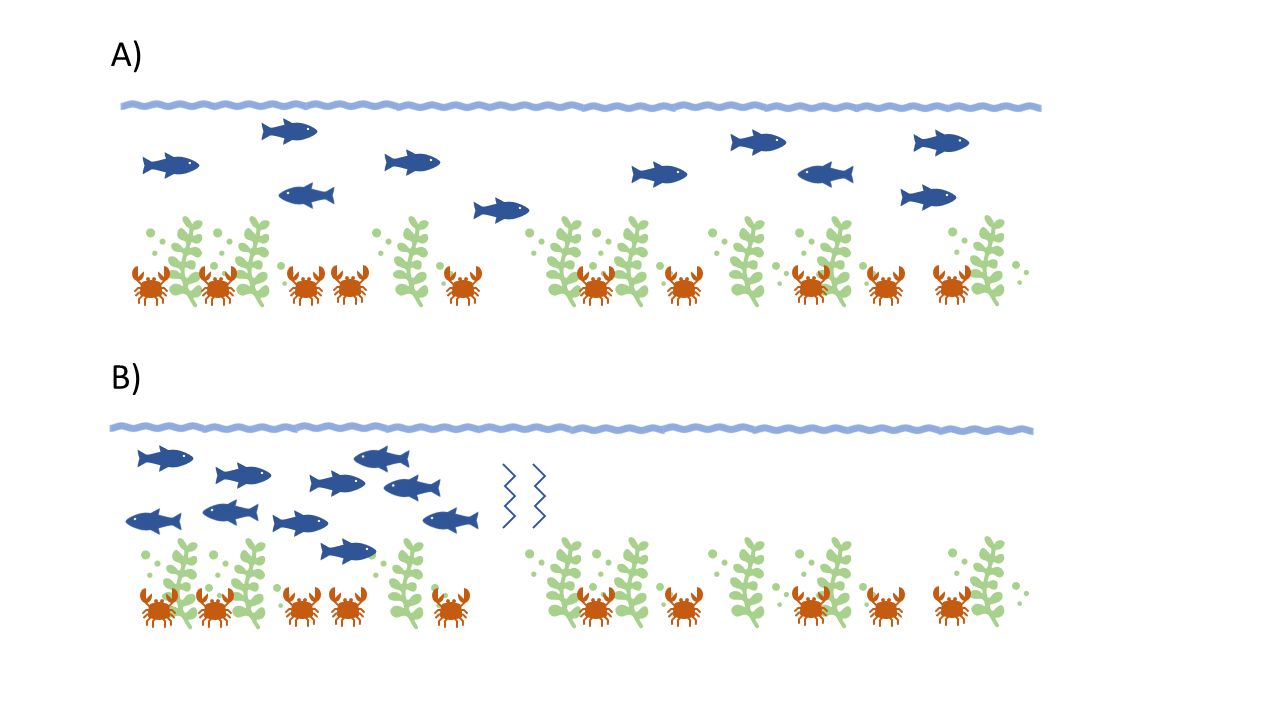
Credit: Gabrielle Love
Recruitment Issues in Florida
There are many things that alter fish recruitment in Florida. Because so many environmental and ecological factors can influence recruitment, it is necessary to understand how the current and predicted future conditions in Florida will continue to affect fish recruitment (and, therefore, entire populations) (Camp et al. 2021). Changes to the natural structures of fish populations or communities (often human-induced) can alter the number of young fish present, or they can change how fish compete for resources during recruitment. Overharvesting or large fish kills decrease reproductive capacity, which can have long-term impacts for recruitment of fish species that do not spawn often. Gulf sturgeon, for example, were overharvested and suffered important nursery habitat loss when humans altered the rivers they spawn in. Because this species takes a very long time to mature (8 years or more), building back a healthy spawning population takes longer, and the population recovers more slowly than would a species that matures quickly and reproduces often (Flowers et al. 2020). Other impacts on populations may include the introduction of non-native fish like lionfish, which can outcompete native species for food, decreasing native recruitment (Albins and Hixon 2008). These types of changes to recruitment can be difficult to manage because the problem may not even be apparent until the damage is already done.
Climate change is already affecting the water conditions in Florida, therefore directly influencing recruitment. Oceanic and coastal water quality changes, such as increased temperatures, decreased pH, increased salinity, and decreased dissolved oxygen, all push the tolerance limits of fish recruits (Robbins and Lisle 2018). Once recruits can no longer tolerate the water conditions, density-dependent effects are irrelevant; fish in intolerable conditions will not survive for long (Sogard 1997). Harmful algal blooms like red tide events have the potential to increase in frequency and severity with climate change, and these extreme events result in massive fish kills (Gilbert and Burford 2017). Red tide can have direct impacts on fish populations by killing spawning adults (reducing the number of larvae produced later) and by killing recruits themselves (Flaherty and Landsberg 2011), as well as indirect impacts on fish by affecting the health of their forage and habitat. Changing water flow conditions (e.g., river modifications for development of water retention; changes in stormwater drainage; increases or decreases in rainfall) will bring further changes to other water conditions and habitats. The way these changes impact recruitment will be highly variable and species-specific, so researchers must monitor them closely to understand how they affect fish. The good news is that water flow can be somewhat controlled with clever water-management strategies, such as dam or reservoir management (Bonvechio and Allen 2005). Other aspects of water quality, however, are not so easy to directly influence, so climate change itself must be considered, and steps should be taken to understand and mitigate for changing water conditions.
Degradation or outright loss of nursery habitats by climate change or other human activities has the potential to significantly reduce the recruitment potential for the species that use them. Many aquatic habitats throughout Florida have become at least somewhat degraded through development and environmental manipulation, including rivers, oyster reefs, and coral reefs (Beck et al. 2011; Hein et al. 2020). For some specific effects of climate change on oysters and their predators, please see FA228. There is a high interest in efforts to restore these crucial nursery habitats in Florida in order to protect the animals that use them. Unfortunately, restoration projects are costly and difficult to execute, and they do not always improve recruitment like they are expected to (Hein et al. 2020). This means it may be less costly to prevent disturbing the habitats in the first place.
Conclusions
A large suite of conditions impact fish recruitment. Some of these conditions affect fish recruitment through density-independent processes, like water quality, flow, and habitat. Some of them impact fish populations through density-dependent processes, driven by competition for things like food and space. Many of these factors can interact with one another, making the recruitment process complex and the degree of recruitment success difficult to predict. Because recruitment is such an important part of the life of a fish, it is important for natural resource managers to preserve the best recruitment conditions possible for Florida’s aquatic communities in order to conserve the natural environment that makes Florida special.
References
Albins, M., and M. Hixon. 2008. “Invasive Indo-Pacific Lionfish Pterois volitans Reduce Recruitment of Atlantic Coral-Reef Fishes.” Marine Ecology Progress Series 367:233–238. https://doi.org/10.3354/meps07620
Anderson, J. T. 1988. “A Review of Size Dependent Survival During Pre-Recruit Stages of Fishes in Relation to Recruitment.” Journal of Northwest Atlantic Fishery Science 8:55–66.
Beck, M. W., R. Brumbaugh, L. Airoldi, A. Carranza, L. Coen, C. Crawford, O. Defeo, et al. 2011. “Oyster Reefs at Risk and Recommendations for Conservation, Restoration, and Management.” BioScience 61 (2): 107–116. https://doi.org/10.1525/bio.2011.61.2.5
Beck, M. W., K. L. Heck, K. W. Able, D. L. Childers, D. B. Eggleston, B. M. Gillanders, B. Halpern, et al. 2001. “The Identification, Conservation, and Management of Estuarine and Marine Nurseries for Fish and Invertebrates.” BioScience 51 (8): 633–641. https://doi.org/10.1641/0006-3568(2001)051[0633:TICAMO]2.0.CO;2
Bonvechio, T. F., and M. S. Allen. 2005. “Relations between Hydrological Variables and Year-Class Strength of Sportfish in Eight Florida Waterbodies.” Hydrobiologia 532 (1–3): 193–207. https://doi.org/10.1007/s10750-004-1388-y
Camp, E., A. B. Collins, R. N. Ahrens, and K. Lorenzen. 2020. “Fish Population Recruitment: What Recruitment Means and Why It Matters FA222. EDIS 2020 (2): 6–6. https://doi.org/10.32473/edis-fa222-2020
Camp, E. V., A. B. Collins, R. N. Ahrens, and K. Lorenzen. 2021. “Fish Population Recruitment 2: Stock Recruit Relationships and Why They Matter For Stock Assessment FA234. EDIS 2021(5). https://doi.org/10.32473/edis-fa234-2021
Doherty, P. J., and D. McB. Williams. 1988. “The Replenishment of Coral Reef Fish Populations. Oceanography and Marine Biology: An Annual Review 26:487–551.
Flaherty, K. E., and J. H. Landsberg. 2011. “Effects of a Persistent Red Tide (Karenia brevis) Bloom on Community Structure and Species-Specific Relative Abundance of Nekton in a Gulf of Mexico Estuary.” Estuaries and Coasts 34 (2): 417–439. https://doi.org/10.1007/s12237-010-9350-x
Flowers, H. J., W. E. Pine, B. T. van Poorten, and E. V. Camp. 2020. “Evaluating Population Recovery Characteristics and Potential Recovery Actions for a Long-Lived Protected Species: A Case History of Gulf Sturgeon in the Apalachicola River.” Marine and Coastal Fisheries 12 (1): 33–49. https://doi.org/10.1002/mcf2.10105
Gilbert, P. M., and M. A. Burford. 2017. “Globally Changing Nutrient Loads and Harmful Algal Blooms: Recent Advances, New Paradigms, and Continuing Challenges.” Oceanography 30 (1): 58–69. https://doi.org/10.5670/oceanog.2017.110
Hein, M. Y., R. Beeden, R. A. Birtles, T. J. Chase, F. Couture, E. Haskin, N. Marshall, et al. 2020. “Effects of Coral Restoration on Fish Communities: Snapshots of Long-Term, Multiregional Responses and Implications for Practice.” Restoration Ecology 28 (5): 1158–1171. https://doi.org/10.1111/rec.13177
Lenihan, H. S., C. H. Peterson, J. E. Byers, J. H. Grabowski, G. W. Thayer, and D. R. Colby. 2001. “Cascading of Habitat Degradation: Oyster Reefs Invaded by Refugee Fishes Escaping Stress.” Ecological Applications 11 (3): 764–782. https://doi.org/10.1890/1051-0761(2001)011[0764:COHDOR]2.0.CO;2
Lorenzen, K., and E. V. Camp. 2019. “Density-Dependence in the Life History of Fishes: When Is a Fish Recruited?” Fisheries Research 217:5–10. https://doi.org/10.1016/j.fishres.2018.09.024
Peterson, C. H., J. H. Grabowski, and S. P. Powers. 2003. “Estimated Enhancement of Fish Production Resulting from Restoring Oyster Reef Habitat: Quantitative Valuation.” Marine Ecology Progress Series 264:249–264. https://doi.org/10.3354/meps264249
Robbins, L. L., and J. T. Lisle. 2018. “Regional Acidification Trends in Florida Shellfish Estuaries: A 20+ Year Look at pH, Oxygen, Temperature, and Salinity.” Estuaries and Coasts 41 (5): 1268–1281. https://doi.org/10.1007/s12237-017-0353-8
Sogard, S. M. 1997. “Size-Selective Mortality in the Juvenile Stage of Teleost Fishes: A Review.” Bulletin of Marine Science 60 (3): 1129–1157.
zu Ermgassen, P. S. E., J. H. Grabowski, J. R. Gair, and S. P. Powers. 2016. “Quantifying Fish and Mobile Invertebrate Production from a Threatened Nursery Habitat.” Journal of Applied Ecology 53 (2): 596–606. https://doi.org/10.1111/1365-2664.12576