Anthracnose on Pepper in Florida
Anthracnose on pepper can be a serious problem for growers in Florida. Widespread outbreaks of the disease have occurred in some seasons on bell and specialty peppers, such as cubanelle and jalapeño. Disease initiated when both the inoculum and ideal weather conditions, such as warm weather and frequent rains, were present. Anthracnose can cause significant damage to mature (red, yellow or orange) and immature fruit (green) resulting in extensive loss of pepper fruit.
Pathogen
Anthracnose is caused by fungi in the genus Colletotrichum, which is a very common group of plant pathogens, causing diseases on numerous plant species worldwide. Identification of Colletotrichum to species is usually based on more than one characteristic, such as morphological (physical appearance) and pathogenicity on host(s), and more recently using molecular analysis. Many species of Colletotrichum infect more than one host and, in addition, more than one species of Colletotrichum may be present on a single host. Both Colletotrichum gloeosporioides and Colletotrichum acutatum cause anthracnose on pepper in Florida and elsewhere (Harp 2008; Lewis-Ivey 2004). Interestingly, C. gloeosporioides was recovered from ripe fruit whereas C. acutatum was identified from the lesions on immature fruit (Harp et al. 2008). The disease on immature fruit caused by C. acutatum has been termed "early anthracnose" to distinguish it from the form of anthracnose caused by C. gloeosporioides occurring on mature fruit.
Symptoms
Colletotrichum is capable of causing disease on virtually all parts of the pepper plant during any stage of plant growth. However, fruit lesions are the most economically important aspect of this disease. Fruit symptoms initially begin as water-soaked lesions that become soft, slightly sunken, and tan. The lesions can cover most of the fruit surface and multiple lesions occur (Figure 1). The surface of the lesion becomes covered with the wet, gelatinous spores from salmon-colored fungal fruiting bodies (acervuli) with numerous black spines (setae) (Figure 2). Concentric rings of the acervuli are common within the fruit spots. In some cases, the lesions are brown, not orange, and then black from the formation of setae and sclerotia (a dark, fungal survival structure) (Figure 3).
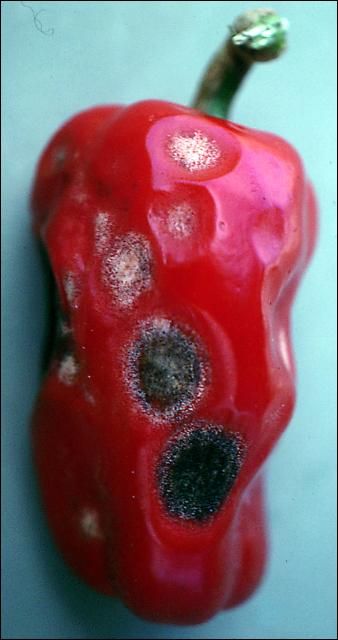

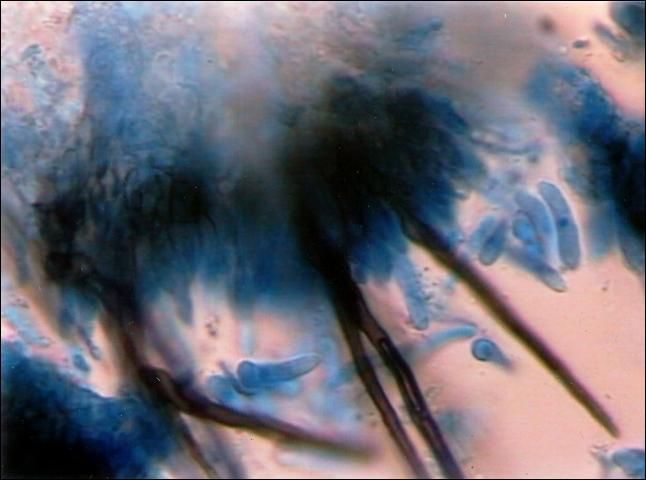
Disease Cycle and Epidemiology
The pathogen survives in and on seeds. Anthracnose is introduced into the field on infected transplants or it can survive between seasons in plant debris or on weed hosts. Alternative hosts have not been fully identified as occurring naturally in the field but probably includes some weeds and other plants in Solanaceae.
Fruit are infected when spores of the fungus or infested debris are rain splashed onto pepper plants. New spores are produced within the infected tissue and then are dispersed to other fruit. Workers may also move spores with equipment or during handling of infected plants.
Infection usually occurs during warm, wet weather. Temperatures around 80°F (27°C) are optimum temperatures for disease development, although infection occurs at both higher and lower temperatures. Severe losses occur during rainy weather because the spores are washed or splashed to other fruit resulting in more infections. If C. gloeosporioides is present, the disease is more likely to develop on mature fruit that is present for a long period on the plant. Anthracnose can occur on both immature and mature fruit.
Control
Control of the disease is through integrated management techniques. The disease should not be introduced on infected plants. Only seeds that are pathogen-free should be planted. Transplants should be kept clean by controlling weeds and solanaceous volunteers around the transplant houses. The field should have good drainage and be free from infected plant debris. If disease was previously present, crops should be rotated away from solanaceous plants for at least two years. Sanitation practices in the field include control of weeds and volunteer peppers plants.
Resistance is available in some varieties of chili peppers but not in bell peppers. For bell pepper production, a cultivar that bear fruit with a shorter ripening period may allow the fruit to escape infection by the fungus. Wounds in fruit from insects or other means should be reduced to the extent possible because wounds provide entry points for Colletotrichum spp. and other pathogens such as bacteria that cause soft rot. Applications of fungicides may be needed to manage the disease. Currently registered fungicides for this disease are listed in the UF/IFAS Vegetable Production Handbook, Pepper Production at https://edis.ifas.ufl.edu/cv130. At the end of the season, the crop debris should be destroyed to reduce survival between crops.
References
Harp T.L., K. Pernezny, M. L. Lewis Ivey, S. A. Miller, P. J. Kuhn, L. and Datnoff. 2008. "The etiology of recent pepper anthracnose outbreaks in Florida." Crop Protection 27: 1380–1384.
Lewis-Ivey M.L., C. Nava-Diaz, and S. A. Miller. 2004. "Identification and management of Colletotrichum acutatum on immature bell peppers." Plant Dis. 88: 1198–1204.


